- Biomedical Research (2014) Volume 25, Issue 4
Analysis of factors causing intracranial infection after endoscopic resection of pituitary tumors by transnasal-sphenoidal approach.
Liqun Zhang1 and Maohua Chen2,*1Department of Otorhinolaryngology, Wenzhou Central Hospital, Wenzhou 325000, China
2Department of Neurosurgery, Wenzhou Central Hospital, Wenzhou 325000, China
Accepted May 05 2014
Abstract
This study analyzed the factors that caused intracranial infection after endoscopic resection of the pituitary tumors by transnasal-sphenoidal approach. A retrospective analysis was performed for 326 patients with resection of pituitary tumor, and multiple Logistic regression analysis was conduced for the relevant factors. Results showed that a total of 32 patients exhibited postoperative infection (9.82%). The infection rates in patients with operation time of > 7 hours, cerebrospinal fluid leakage, diabetes mellitus, as well as those receiving corticosteroids, preoperative use of antibiotics and postoperative use of antibiotics for > 7 days were 15.56%, 15.22%, 13.75%, 9.94%, 10.00% and 10.47%, respectively. The longer operative time, the accompanying cerebrospinal fluid leakage and diabetes mellitus were the infection-related factors after endoscopic resection of pituitary tumors by transnasal-sphenoidal approach (P <0.05), while the preoperative use of corticosteroids and antibiotics and the postoperative use of antibiotics had no beneficial effects on the postoperative accompanying infection (P> 0.05). Thus, intracranial infection after endoscopic resection of pituitary tumors by transnasal-sphenoidal approach seems to be affected by many factors. Further survey and analysis with larger sample size are still needed.
Keywords
Intracranial, infection, endospic, pituitary, logistic regression, transnasal-sphenoidal
Introduction
Endoscopic resection of pituitary tumors by transnasal-sphenoidal approach has been wildly used in clinic [1]. However, with the drug-resistant strains coming out constantly and the abuse of the broad-spectrum antibiotic in clinic, intracranial infection has attracted more and more attention of medical staff. In this study, a retrospective analysis of factors related to intracranial infection after endoscopic resection of pituitary tumors through the nasal sphenoid was adopted to discover risk factors for intracranial infection after endoscopic resection of pituitary tumors and provide the clinical basis for better control of nosocomial infection after endoscopic resection of pituitary tumors by transnasal-sphenoidal approach.
Materials and Methods
Clinical data
Three hundred and twenty six (326) hospitalized patients in our neurosurgical department from January 2007 to April 2011 were chosen, including 177 males and 149 females (mean age: 33.28±6.78 [15-65]; course of disease: 2 months to 5.0 years). The major clinical manifestations include headache, vision loss and visual field defect as well as menelipsis, galactorrhea, sexual debility and erectile dysfunction like in diabetes insipidus. CT and MRI examinations of head showed 34 cases of microadenoma (13.43%, D (diameter) < 1 cm), 247 cases of small and medium-sized adenoma (75.77%, 1 cm<D<3 cm) and 45 cases of macroadenoma (13.8%, D>3 cm). CT examination also included axial and coronal CT scan of nasal cavity and paranasal sinus to know the internal structure of nasal cavity and gasification degree of sphenoid sinus. Postoperative pathological examination confirmed 175 patients with prolactin (PRL) adenoma (53.68%), 45 patients with growth hormone (GH) adenoma (13.80%), 36 patients with adrenocorticotrophic hormone (ACTH) adenoma (11.04%), and 70 patients with nonfunctioning adenoma (21.47%).
Operative procedures
After induction of general anesthesia and endotracheal intubation, the horizontal position was adopted, and the head of the patient was extended back by 30° and flexed by 20° toward the right side to be fixed in a semireclining position. About 1.5 cm longitudinal incision of septum mucosa was made along the leading edge of middle turbinate by electrocoagulation using nasal cavity approach. The septum mucosa was stripped to the anterior wall of the sphenoid sinus with an aspirator, and part of the bony nasal septum was broken; meanwhile, the contralateral septum mucosa was pushed to the anterior wall of the sphenoid sinus and retracted with an aspirator. Partial bony nasal septum was removed by the pith nucleus clamp and the mucosa was stretched to expose the anterior wall of the sphenoid sinus. The anterior wall of sphenoid sinus was opened with a pith nucleus clamp to enter sphenoid sinus, thus exposing the sellar floor. The mucosa of the sellar floor was given firing shrinkage and the sellar floor was opened with an abrasion drill.
A cross cut on the dura mater was made with bipolar coagulation to enter the sellar area, and tumor forceps, curet and aspirator were used to remove the intrasellar tumor piece by piece under an endoscope. The tumor and the surrounding structures were observed using 0o and 30o endoscopes to remove the latent tumors.
Finally, hematischesis was performed, and the tumor cavity was filled with gelatin sponge. The sellar floor was repaired if there was no residual cerebrospinal fluid.
Statistical analysis
SPSS 17.0 was used for statistical analysis of the data. Multiple Logistic regression analysis was performed with infection rates as dependent variable and other factors as independent variables.
Results
Impact of different factors on the postoperative infection rate
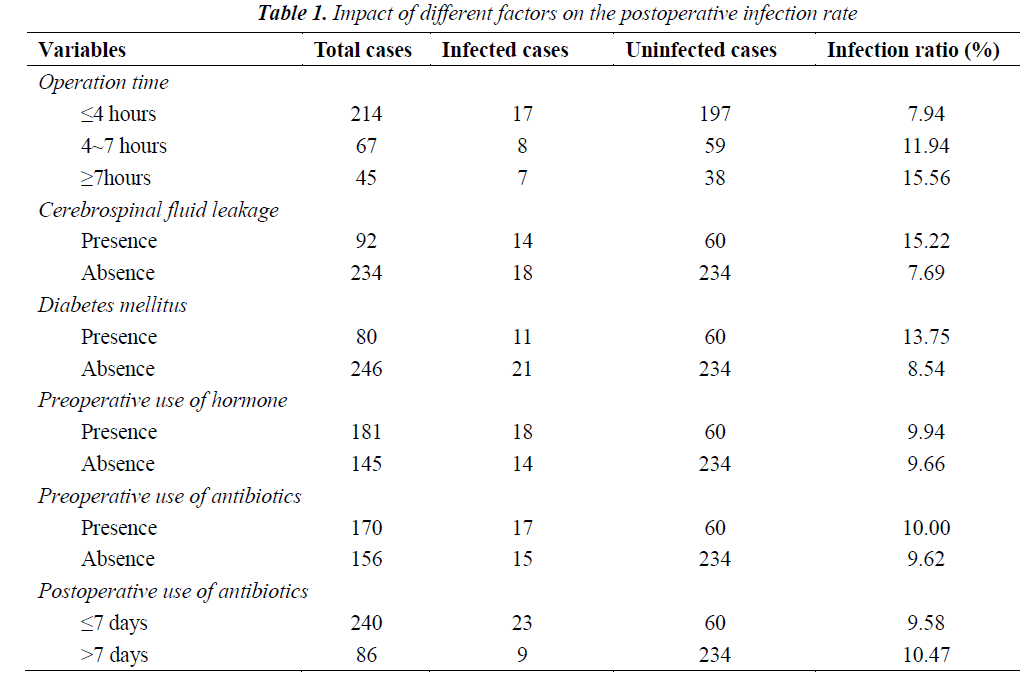
Impacts of different factors on the postoperative infection rate were summarized in Table 1. As shown in Table 1, 32 of 326 patients presented postoperative infections (9.82%). The infection rates in patients with operation time of > 7 hours, cerebrospinal fluid leakage and diabetes, as well as those receiving hormone, preoperative use of antibiotics and postoperative use of antibiotics for > 7 days were 15.56%, 15.22%, 13.75%, 9.94%, 10.00% and 10.47%, respectively.
Multi-factor analysis on the infection-related factors
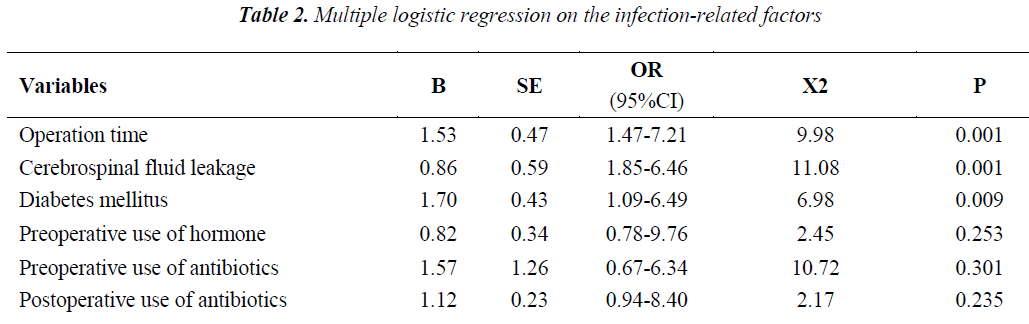
The results of multiple Logistic regression analysis on the infection-related factors can be seen in Table 2. As shown in Table 2, the longer operative time and the accompanying cerebrospinal fluid leakage and diabetes mellitus were the infection-related factors after endoscopic resection of pituitary tumors by transnasal- sphenoidal approach (P < 0.05), while the preoperative use of hormone and antibiotics and the postoperative use of antibiotics had no obvious effect on the postoperative accompanying infection (P>0.05).
Discussion
Intracranial infection after endoscopic resection of pituitary tumors by transnasal-sphenoidal approach is relatively complicated and can be affected by multiple factors. Due to protections of the blood brain barrier (BBB), meninges, skull, scalp and other tissues, the incidence of intracranial infection is relatively lower. However, neurosurgery can make brain tissues of BBB connect with the outside environment, thus increasing the incidence of intracranial infection [2]. An increased postoperative infection is also gradually resulted due to increasing drug-resistant strain and the abuse of the antibiotics.
The results of intracranial infection and the reported infection ratio differ greatly, generally ranging from 1.52% to 15.51% [3-5], and some even as high as 27.59% [6]. Among the 326 patients in our study, 32 patients were infected (9.82%). which was similar to the data presented in literatures. The infection ratio is easily affected by multiple factors such as clinical skill of the surgeon, criteria for infection diagnosis and social factors, but the opinions of factors easily leading to infection are much the same.
The neurosurgery time is influenced by the complexity of operation, surgical techniques of the surgeon and extent of tissue damage. The longer operation time and operative field exposure time bring the tissues more in touch with air, hands of surgeons and other objects, thus resulting in a higher infection ratio after operation. In our study, the patients of the group with a longer operation time suffered a higher intracranial infection ratio. Operation time is an affecting factor of the intracranial infection ratio after operation through the statistical analysis. The cerebrospinal fluid leakage after neurosurgery is a high risk factor of intracranial infection after operation, which greatly increases the infection rate. In this study, the infection rate of patients with cerebrospinal fluid leakage reached up to 15.22%. It has been reported that the infection rate of patients with cerebrospinal fluid leakage is 16.7% [7], which is close to our results. Thus, it could be suggested that cerebrospinal fluid leakage is an important risk factor of intracranial infection after operation, which is also proved through our Logistic regression analysis. In patients with diabetes mellitus, the postoperative infection occurs more easily because vasculopathy can cause a decrease in blood flow, and high blood glucose is beneficial to bacteria growth and reproduction, can suppress the phagocytic ability of leukocytes and make the tissue function of the patients decrease to respond to a lower amount of antibiotics. This was also analyzed by Tsou TP et al. [8].
In the early stage, a significant cerebral protective effect can be caused after a high dose of glucocorticoids, which brought a new understanding of hormone application to people [9]. In recent years, we also apply hormone before operation (especially in patients with pituitary adenoma) due to its influence on the neuroendocrine function, thus reducing stress responses [10]. Our results showed that the infection ratio of patients receiving hormone before surgery was 9.94%, while the ratio in patients who didn’t receive hormone before surgery was 9.66%. The results were not significantly different (P>0.05).
The application of hormone did not increase the incidence of nosocomial infection after surgery for pituitary tumors, and Logistic regression analysis showed that hormone did not have an obvious impact on the postoperative infection (P>0.05). In our study, it was also found that the intracranial infection rates in patients with preoperative use of antibiotics and those without preoperative use of antibiotics were 10.00% and 9.60%, respectively. Regression analysis shows that the preoperative use of antibiotics can reduce the incidence of intracranial infection without a correlation. The results after preoperative application of antibiotics for 7 days were the same as that after preoperative application of antibiotics for 7 days. Therefore, long-term administra tion of antibiotics before or after operation is unnecessary.
Endoscopic resection of the pituitary tumors by transnasal- sphenoidal approach is a proficient operation. However, further investigation is needed to ensure that the technique becomes apparently free from infection.
References
- Fatemi N, Dusick JR, de Paiva Neto MA, et al. The endonasal microscopic approach for pituitary adenomas and other parasellar tumors: a 10-year experience. Neurosurgery 2008; 63: 244-256.
- Bisceglia M, Simeone A, Forlano R, et al. Fatal systemic venous air embolism during endoscopic retrograde cholangiopancreatography. Adv Anat Pathol 2009; 16: 255-262.
- Korinek AM, Baugnon T, Golmard JL, et al. Risk factors for adult nosocomial meningitis after craniotomy: role of antibiotic prophylaxis. Neurosurg- ery 2006; 59: 126-133.
- Korinek AM, Golmard JL, Elcheick A, et al. Risk factors for neurosurgical site infections after craniotomy: a critical reappraisal of antibiotic prophyla- xis on 4,578 patients. Br J Neurosurg 2005; 19:155-162.
- Brown SM, Anand VK, Tabaee A, et al. Role of perioperative antibiotics in endoscopic skull base surgery. Laryngoscope 2007; 117: 1528-1532.
- K, Kraoul L, Alyousef L, et al. [The role of biology in the diagnosis of cerebrospinal fluid leaks]. Ann Biol Clin (Paris) 2009; 67: 141-151.
- Tsou TP, Lee PI, Lu CY, et al. Microbiology and epidemiology of brain abscess and subdural empyema in a medical center: a 10-year experience. J Microbiol Immunol Infect 2009; 42: 405-412.
- Meltser I, Canlon B. Protecting the auditory system with glucocorticoids. Hear Res 2011; 281: 47-55.
- Doi M, Sugiyama T, Izumiyama H, et al. Clinical features and management of ectopic ACTH syndrome at a single institute in Japan. Endocr J 2010; 57: 1061-1069.
- Jin GM, Dong YM, Yu AR, et al. Meta-analysis of the epidemiological investigation on intracranial infection after craniotomy. Chin J Clin Neurosurg 2007; 12: 149-151.