Rapid Communication - Biomedical Research (2017) Volume 28, Issue 11
Altered expression of sex hormone receptors in keratoconus corneas
Hongbo Yin1, Chang Luo2, Yan Tian1 and Yingping Deng1*
1Department of Ophthalmology, West China Hospital, Sichuan University, 37 Guo Xue Xiang, Chengdu, Sichuan, PR China
2Department of Ophthalmology, Tianfu New Area People's Hospital, 179 Zhengbei Street, Tianfu New Area, Chengdu, Sichuan, PR China
- *Corresponding Author:
- Yingping Deng
Department of Ophthalmology
West China Hospital
Sichuan University, PR China
Accepted date: April 5, 2017
Abstract
Introduction: Keratoconus is an ectasia that results in corneal thinning in central and paracentral areas of affected corneas. This disease occurs most frequently in young males, underlying a potential role of sex hormones in its pathogenesis. By examining the expression of sex hormone receptors, this study aimed to reveal potential roles of the sex hormone receptors in keratoconus pathogenesis.
Methods: Keratoconus and non-keratoconus control corneas were collected from patients volunteered in the West China Hospital. 16 corneas from male patients diagnosed with keratoconus and 9 control corneas from non-keratoconus males were subjected to immunohistochemical staining. The expression of 4 sex hormone receptors, progesterone, androgen and estrogen receptor alpha and estrogen receptor beta was evaluated using their specific antibodies.
Results: Compared with the healthy corneas, the progesterone receptor showed significantly lower expression (P=0.01), while androgen receptor expression was significantly higher in keratoconus corneas (p=0.017). No significant difference in estrogen receptor alpha (P=0.978) and estrogen receptor beta (P=0.276) was detected between keratoconus and control corneas. No significant difference in estrogen receptor alpha and estrogen receptor beta was detected between keratoconus and control corneas.
Conclusions: The reduction of progesterone receptor expression and increment of androgen receptor expression in keratoconus corneas support that sex hormone receptors are involved in the development of keratoconus.
Keywords
Keratoconus, Progesterone receptor, Androgen receptor, Estrogen receptor alpha, Estrogen receptor beta
Introduction
Gender and age predilections have been found in many ocular conditions, which support a potential role of sex hormones in maintaining the eye health [1]. Eye disorders such as agerelated Macular Degeneration (AMD), idiopathic full-thickness macular hole, glaucoma, cataract, and even the dry eye have been constantly found associated with gender or age in many studies [2-8]. Hormone replacement therapy has been reported to ameliorate some of the eye disorders [9].
Keratoconus is a non-inflammatory disorder that results in thinning in central and paracentral areas of affected corneas, which consequently leads to astigmatism [10]. The pathogenesis of keratoconus is still elusive. Gender and age related differences are observed in keratoconus patients. During the past decade, studies have revealed a higher occurring frequency of keratoconus in males than in females and the onset of this disorder begin mainly in adolescence [11-15]. Sex hormone receptors have been found to be expressed in corneas where they regulate the corneal functions [16-18]. We hypothesized that there was a connection between keratoconus and sex hormone receptors. To examine this hypothesis, we performed Immunohistochemistry (IHC) analysis against four sex steroid receptors, Progesterone Receptor (PR), Androgen Receptor (AR), Estrogen Receptor alpha (ERα), Estrogen Receptor beta (ERβ) with keratoconus and normal corneas collected from West China Hospital. Our results demonstrated that the expression of progesterone receptor was downregulated and the androgen receptor expression was upregulated in keratoconus corneas, while ERα and ERβ showed similar expression levels in keratoconus and normal groups.
Methods
Cornea collection
The human corneas were collected from donors who volunteered their corneas and gave consent to use their corneas for this research. The keratoconus corneas were harvested during penetrating keratoplasty. While the non-keratoconus control corneas were obtained from ocular trauma victims with intact corneas who underwent ocular enucleation. All the patients were operated under systemic anesthesia.
In penetrating keratoplasty, the recipient cornea was trephined with a Hessburg-Barron suction trephine. The anterior chamber was then entered with a disposable sharp blade followed by removing the recipient corneal button with the curved corneal scissors. In ocular enucleation, the cornea with 2 mm sclera rim was cut with the curved corneal scissors. And then a 7.5 mm corneal button was punched out using the Barron donor punch. The samples were fixed in formalin immediately and sent to Department of Pathology for paraffin embedding. This protocol was approved by the Institutional Review Board of West China Hospital, Sichuan University.
Immunohistochemistry (IHC). Immunohistochemical staining was performed on all specimens with a single application for each antibody to minimize procedural variations. All the human cornea tissue slides were sectioned with thickness of 2 mm to 3 mm using a microtome. Slides were deparaffinized in xylene and hydrated through graded alcohols. For antigen retrieval, sections were autoclaved in citric acid buffer (pH 6.0) at 121°C for 10 min. The sections were treated with 0.3% hydrogen peroxide in methanol for 30 min at room temperature to block endogenous peroxidase activity and incubated with 5% Bovine Serum Albumin (BSA) in 50 mM Tris-buffered saline (pH 7.4) containing 0.05% Triton X-100 for 1 h at room temperature to block non-specific protein binding site. The sections were then incubated with the primary antibodies at specific dilutions overnight. The AR and PR antibodies (Rabbit polyclonal, GeneTex USA) were diluted at 1:100 and the ERα and ERβ antibodies (Rabbit polyclonal, GeneTex USA) were diluted at 1:50 and 1:400 respectively. The staining was visualized using EnVision System-HRP (DAB Dako Cytomation, USA). Harris’s hematoxylin was applied for 15 sec and then washed with distilled water. The sections were gradually dehydrated with 50%-100% ethanol and xylol. Coverslips were affixed using Entellan for analysis under an optical microscope.
Quantification of IHC
All cases were evaluated by two persons in a double blind manner. Five view fields were chosen randomly at 40 × 10 magnification and at least 100 cells were required for scoring in each field. Cases were scored as 1, 2, 3 or 4 corresponding to positive nuclei rates of 0-25%, 25-50%, 50-75% and 75-100% respectively. Scores from 1-4 for staining intensity were also evaluated for each field. Scores for positive nuclei rates and staining intensity were averaged to produce a final score for each field. The score for one slide is obtained by averaging the scores of five independent fields.
Data analysis
All the analysis was performed using SPSS 11.5. To assess statistical significance, we used the Mann-Whitney U test to compare keratoconus group with the control group and P value<0.05 was considered statistically significant.
Results
Age distribution of the corneas used for IHC analysis
Sixteen keratoconus and 9 non-keratoconus corneas were collected from male donators. To avoid the possible bias introduced by ages, we first compared the age distributions of our samples. Samples were allocated into four age groups: <20, 21-30, 31-40, >41. Evaluation using Mann-Whitney U indicated there was no significant difference in the age distribution between the keratoconus and control groups (P=0.76).
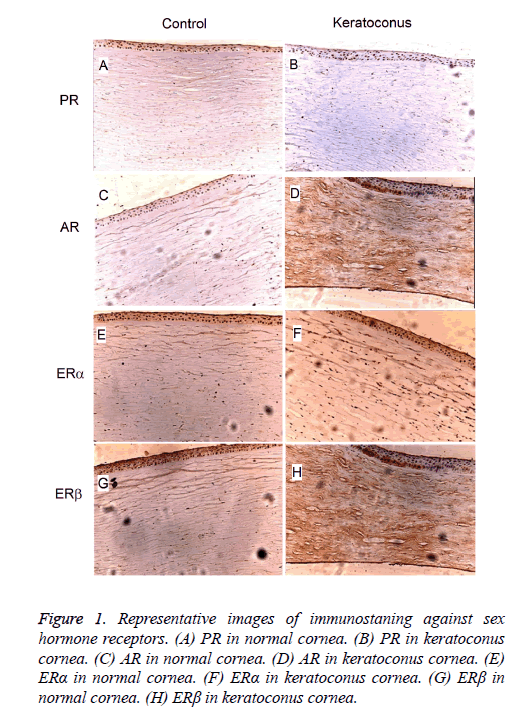
Sex hormone receptors were expressed in the corneas. Four types of sex hormone receptors including Progesterone Receptor (PR), Androgen Receptor (AR), Estrogen Receptor alpha (ERα) and Estrogen Receptor beta (ERβ) were subjected to the IHC staining. Expression of these receptors was detected in the epithelium layer, stroma as well as the endothelium of the cornea (Figure 1). Consistent with previous studies [17,18], we found the existence of sex hormone receptors in both the keratoconus and normal corneas, suggesting a potential role of sex hormones in physiology or pathology of the cornea.
Figure 1: Representative images of immunostaning against sex hormone receptors. (A) PR in normal cornea. (B) PR in keratoconus cornea. (C) AR in normal cornea. (D) AR in keratoconus cornea. (E) ERα in normal cornea. (F) ERα in keratoconus cornea. (G) ERβ in normal cornea. (H) ERβ in keratoconus cornea.
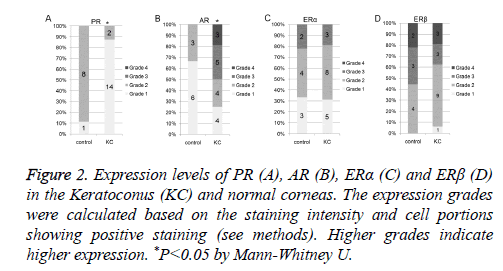
PR expression was reduced in keratoconus corneas. We employed a scoring system containing 1-4 grades to quantify the protein expression level in IHC staining (see methods). Higher grades indicated higher expression levels. As shown in Figure 2A, there were 87.5% (14 out of 16) of keratoconus corneas showing PR expression level of grade 1 and 12.5% (2 out of 16) of them showing PR expression level of grade 2. In control group, 11.1% of the corneas (1 out of 9) showed expression level of grade 1 and the rest 88.9% showed expression level of grade 2. Evaluation using Mann-Whitney U test indicated that the expression level of PR in keratoconus corneas was significantly lower than that in the normal corneas (P=0.01).
Figure 2: Expression levels of PR (A), AR (B), ERα (C) and ERβ (D) in the Keratoconus (KC) and normal corneas. The expression grades were calculated based on the staining intensity and cell portions showing positive staining (see methods). Higher grades indicate higher expression. *P<0.05 by Mann-Whitney U.
AR expression was increased in keratoconus corneas. In keratoconus group, the percentages of AR expression at grade 1, 2, 3, 4 were 25.0% (4 out of 16), 25.0% (4 out of 16), 31.3% (5 out of 16) and 18.8% (3 out of 16) respectively (Figure 2B). In control corneas, the AR showed expression at either grade 1 (66.7%, 6 out of 9) or grade 2 (33.3%, 3 out of 9) (Figure 2B). Compared with control corneas, AR expression in keratoconus corneas was significantly increased (Mann-Whitney U P=0.017).
Expression levels of ERα and ERβ was not significantly altered. Compared with the control corneas, the expression grades of ERα and ERβ in keratoconus corneas showed very similar distribution (Figures 2C and 2D). We found no statistical difference in either ERα expression (Mann-Whitney U, P=0.978) or ERβ expression (Mann-Whitney U, P=0.276) between keratoconus and control corneas.
Discussion
In the current study, we found that the expression of sex hormone receptor PR was downregulated while expression of AR was upregulated in keratoconus corneas. The upregulation of AR in keratoconus corneas is consistent with the finding that conical change in cornea is androgen dependent in the mouse model of keratoconus [19]. Keratoconus occurs more frequently in males who have higher levels of PR and lower levels of AR in the body than the females [11-13,20]. Together with these epidemiologic results, our studies support that PR and AR might be involved in the pathogenesis of keratoconus. However, an alternative explanation that alteration of the sex hormone receptor expression is manifestations of keratoconus is also plausible.
Corneal biomechanical parameters and central thickness are reported to change along with the oscillations of estrogen level during the menstrual cycle in female [21-23], and hormone replacement therapy are found to increase corneal thickness [24]. The female hormone estrogen has been widely reported to modulate the tumor microenvironment [25]. Recently, female hormone estrogen is reported to be able to inhibit collagen degradation [26], which provides a plausible explanation for the fact that keratoconus occurs less frequently in women. Though our results suggested that the expression of the ERα and ERβ were not significantly altered in the keratoconus corneas, it is still to be determined if their activity is changed in keratoconus corneas.
In conclusion, the expression of PR was downregulated and expression of AR was upregulated in keratoconus, which might contribute to the variations by gender and age in keratoconus occurrence. Though further studies are required to determine the causal relationship between sex hormone receptors and keratoconus, the hypothesis that sex hormones receptors are involved in the pathogenesis of keratoconus is in accordance with the sex and age predilection of keratoconus.
Acknowledgment
This study was supported by China National Natural Science (Grant No. 31200711).
Conflict of Interest
The authors declare no conflict of interest.
References
- Wagner H, Fink BA, Zadnik K. Sex- and gender-based differences in healthy and diseased eyes. Optometry 2008; 79: 636-652.
- Rudnicka AR, Jarrar Z, Wormald R, Cook DG, Fletcher A. Age and gender variations in age-related macular degeneration prevalence in populations of European ancestry: a meta-analysis. Ophthalmology 2012; 119: 571-580.
- Dynlacht JR, Tyree C, Valluri S, DesRosiers C, Caperell-Grant A. Effect of estrogen on radiation-induced cataractogenesis. Radiat Res 2006; 165: 9-15.
- Colitz CM, Sugimoto Y, Lu P, Barden CA, Thomas-Ahner J. ERalpha increases expression and interacts with TERT in cataractous canine lens epithelial cells. Mol Vis 2009; 15: 2259-2267.
- Pasquale LR, Kang JH. Female reproductive factors and primary open-angle glaucoma in the Nurse Health Study. Eye (Lond) 2011; 25: 633-641.
- Mitchell P, Leung H, Wang JJ, Rochtchina E, Lee AJ. Retinal vessel diameter and open-angle glaucoma: the Blue Mountains Eye Study. Ophthalmology 2005; 112: 245-250.
- Auw-Haedrich C, Feltgen N. Estrogen receptor expression in meibomian glands and its correlation with age and dry-eye parameters. Graefes Arch Clin Exp Ophthalmol 2003; 241: 705-709.
- Krenzer KL, Dana MR, Ullman MD, Cermak JM, Tolls DB. Effect of androgen deficiency on the human meibomian gland and ocular surface. J Clin Endocrinol Metab 2000; 85: 4874-4882.
- Guaschino S, Grimaldi E, Sartore A, Mugittu R, Mangino F. Visual function in menopause: the role of hormone replacement therapy. Menopause 2003; 10: 53-57.
- Rosen ES. Keratoconus. J Cataract Refract Surg 2012; 38: 927-928.
- Owens H, Gamble G. A profile of keratoconus in New Zealand. Cornea 2003; 22: 122-125.
- Ertan A, Muftuoglu O. Keratoconus clinical findings according to different age and gender groups. Cornea 2008; 27: 1109-1113.
- Fink BA, Wagner H, Steger-May K, Rosenstiel C, Roediger T. Differences in keratoconus as a function of gender. Am J Ophthalmol 2005; 140: 459-468.
- Sharma R, Titiyal JS, Prakash G, Sharma N, Tandon R. Clinical profile and risk factors for keratoplasty and development of hydrops in north Indian patients with keratoconus. Cornea 2009; 28: 367-370.
- Szczotka LB, Barr JT, Zadnik K. A summary of the findings from the Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study. CLEK Study Group. Optometry 2001; 72: 574-584.
- Ogueta SB, Schwartz SD, Yamashita CK, Farber DB. Estrogen receptor in the human eye: influence of gender and age on gene expression. Invest Ophthalmol Vis Sci 1999; 40: 1906-1911.
- Wickham LA, Gao J, Toda I, Rocha EM, Ono M. Identification of androgen, estrogen and progesterone receptor mRNAs in the eye. Acta Ophthalmol Scand 2000; 78: 146-153.
- Gupta PD, Johar K, Nagpal K, Vasavada AR. Sex hormone receptors in the human eye. Surv Ophthalmol 2005; 50: 274-284.
- Tachibana M, Adachi W, Kinoshita S, Kobayashi Y, Honma Y. Androgen-dependent hereditary mouse keratoconus: linkage to an MHC region. Invest Ophthalmol Vis Sci 2002; 43: 51-57.
- Fink BA, Sinnott LT, Wagner H, Friedman C, Zadnik K, CLEK Study Group. The influence of gender and hormone status on the severity and progression of keratoconus. Cornea 2010; 29: 65-72.
- Giuffrc G, Di Rosa L, Fiorino F, Bubella DM, Lodato G. Variations in central corneal thickness during the menstrual cycle in women. Cornea 2007; 26: 144-146.
- Goldich Y, Barkana Y, Pras E, Fish A, Mandel Y. Variations in corneal biomechanical parameters and central corneal thickness during the menstrual cycle. J Cataract Refract Surg 2011; 37: 1507-1511.
- Leach NE, Wallis NE, Lothringer LL, Olson JA. Corneal hydration changes during the normal menstrual cycle-a preliminary study. J Reprod Med 1971; 6: 201-204.
- Affinito P, Di Spiezio Sardo A, Di Carlo C, Sammartino A, Tommaselli GA. Effects of hormone replacement therapy on ocular function in postmenopause. Menopause 2003; 10: 482-487.
- Hima S, Sreeja S. Modulatory role of 17β-estradiol in the tumor microenvironment of thyroid cancer. IUBMB Life 2016; 68: 85-96.
- Zhou H, Kimura K, Orita T, Nishida T, Sonoda KH. Inhibition by female sex hormones of collagen degradation by corneal fibroblasts. Mol Vis 2011; 17: 3415-3422.