- Biomedical Research (2015) Volume 26, Issue 1
A comparative study of excretion of the components after oral administration of pure baicalin radix scutellariae and scutellariae-paeoniae couple extracts to normal and ulcerative colitis rats.
Wei Liu1,2, Shuai Zhang1, Xiao-huan Fu1, Hong-juan Li1, Wei Xiao1*, Zhen-Qiu Zhang2*1Jiangsu Kanion Pharmaceutical CO., LTD. No. 58 Haichang Road, Sinpo District of Lianyungang, Jiangsu province, 222001, China
2Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian Liaoning province, 116600, China
- *Corresponding Author:
- Wei Xiao
Jiangsu Kanion Pharmaceutical CO
LTD. No. 58 Haichang Road Sinpo
District of Lianyungang Jiangsu province
China. 222001
Zhang Zhen-Qiu
College of Pharmacy Liaoning
University of Traditional Chinese Medicine
77 Shengming 1 Road
DD Port Dalian, 116600, China.
Accepted August 24 2014
Abstract
Radix scutellariae and Scutellariae-Paeoniae couple extracts are the well-known Traditional Chinese Medicine (TCM) to treat ulcerative colitis. Baicalin, wogonoside, baicalein and wogonin are the main effective ingredients in Radix scutellariae. This study was carried out to investigate the excretion profiles of baicalin, wogonoside, baicalein and wogonin in urine and feces after oral administration of pure baicalin, Radix scutellariae and Scutellariae- Paeoniae couple extracts to rats with ulcerative colitis (UC) and to compare the different profiles of urine with normal rats. The levels of the four flavones in urine and feces were measured by a rapid and sensitive high-performance liquid chromatography (HPLC) method. All the rats were divided randomly into two groups (ulcerative colitis and normal groups). Each group contained three subgroups: pure baicalin, Radix scutellariae and Scutellariae- Paeoniae couple subgroup. Each group received oral administration of pure baicalin, Radix Scutellariae and Scutellariae-Paeoniae couple extracts; which contained nearly the same amounts of baicalin at a dosage of 200 mg/kg. All the four flavones occurred in the rat urine and feces, but the general levels of baicalin and wogonoside were predominant in urine and baicalein and wogonin were predominant in feces. The cumulative excretion quantities of these flavones in rat urine or feces were significantly different among these groups (P < 0.05). Meanwhile, the cumulative excretion quantities of these flavones in UC rat urine were less than that in normal rat. Conclusion: Our results suggest that the combination of Radix Scutellariae and Radix Paeoniae Alba might afford higher absorption and excretion, and that the rate of utilization of pure baicalin, Radix scutellariae or Scutellariae- Paeoniae couple is increased in UC.
Keywords
Flavones; Excretion; Urine; Feces.
Introduction
The use of natural products as herbal medicines has increased steadily over the last decade [1]. Moreover, approximately 80% of the population in Asian countries uses herbal medicines for promoting health and managing common maladies such as cold, inflammation, pain to more serious ones such as heart diseases, liver cirrhosis, diabetes and central nervous system disease [2,3]. Moreover, patients with chronic diseases that are likely to be treated with multiple drugs, use herbal medicines more frequently, thereby increasing the need to study the pharmacokinetics (absorption, distribution, metabolism and excretion) of herbal medicines and drug-drug interactions.
UC is a worldwide, chronic, idiopathic, inflammatory bowel disease (IBD) of the rectal and colonic mucosa, and impacts negatively on the quality of life [4-6]. Thus, the development of a more effective therapy to treat UC disease has become imminent. Huangqing-Tang decoction, described by Zhang Zhongjing (150 to 219 A.D., in Chinese Eastern Han Dynasty), has been used for the treatment of UC for thousands of years. Radix Scutellariae and Radix Paeoniae Alba are the key ingredient herbs. The combination of Radix Scutellariae and Radix Paeoniae Alba (called Scutellariae-Paeoniae couple) renders heat-clearing, anti-inflammatory, anti-diarrheal and antinociceptive effects. Apparently, it is essential to study the synergistic interaction of Radix Scutellariae and Radix Paeoniae Alba for elucidating the substantial foundation of Huangqing-Tang.
The dried root of Scutellaria Radix (commonly called Huangqin) is widely used in TCM. The main components of Scutellariae Radix are baicalin (BG), wogonoside (WG), baicalein (B) and wogonin (W) Fig.1. Scutellariae Radix and its major flavonoids possess multiple biological and pharmacological effects, including anti-inflammation, anti-viral, anti-tumor, anti-proliferative and anti-bacterial, etc.[7-9]. To date, the pharmacokinetic (PK) profiles of BG and WG in plasma, brain and eyes of rats and rabbits have been reported [10-13]. From literature we know that after oral administration of baicalein plasma levels of baicalein were negligible. The glucuronides/sulfates of baicalein were predominant in the plasma [14-17]. There are various case reports on herb–drug interactions or herb– herb interactions in vivo in recent years, and also some reports on the influence of disease condition on the pharmacokinetic characteristics of drugs [18-20].
In our laboratory, an experimental model of UC has been established in male Sprague-Dawley(SD) rats using Trinitro- benzene-sulfonic acid TNBS [21-23]. Recently, we have reported the pharmacokinetics and tissue distribution profiles of baicalin, wogonoside, baicalein and wogonin after oral administration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae couple extracts to normal rats, and compared the pharmacokinetics with UC rats (personal communication) [24]. However, we have not reported the urine or feces excretion profiles of the four flavones. Baicalin, wogonoside, baicalein and wogonin are all part of or some byproducts of metabolism after oral administration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae couple extracts to normal and UC rats. For this reason, this study has been undertaken to explore potential differences in urine or feces excretions among the pure baicalin group, Radix Scutellariae group and Scutellariae-Paeoniae couple group and at the same time compare the similarities and differences of urine excretion between UC rats and normal rats.
Material and Methods
Chemicals
Radix scutellariae and Radix paeoniae alba were purchased from Bozhou Medicine Company (Anhui, China.) and authenticated by Prof. Feng Li from the College of Pharmacy, University of Traditional Chinese Medicine (Liaoning, China). Pure baicalin was obtained from Prof. ZhenQiu Zhang (Dept. of Chemistry in our institute, Liaoning University of TCM). Wogonin was purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). The internal standard (IS) arctigenin was supplied by Prof. DeQiang Dou (Department of Phytochemistry in our institute). Baicalein was purchased from Tianjin Yifang Science & Technology Co. Ltd. (Tianjin, China). Wogonoside was purchased from Shanghai Ronghe Science &Technology Co. Ltd. (Shanghai, China). Trinitrobenzene- sulfonic acid was obtained from Shanghai Jinmai Co. Ltd. Shanghai China. The solvents used for chromatographic analysis were HPLC grade and were purchased from Fisher Company Inc., USA. Deionized water was prepared in a Mill-Q academic water purification system (Millipore, Bedford, MA, USA). All the other reagents were of analytical grade and provided by Kermel Chemical Co. (Tianjin, China).
Apparatus and chromatography
The concentrations of the four flavonoids in plasma were assayed using reverse-phase high performance liquid chromatography (Agilent 1100 series) equipped with a variable wavelength UV detector and pump (Agilent model G1314A VWD). Separation was accomplished on an Eclipsel XDB-C18 column (250 mm × 4.6 mm, 5 μm particle size). Mobile phase A was acetonitrile and phase B was 0.1% (v/v) phosphoric acid aqueous solution. The elution was performed using a linear gradient of (urine:16-16% B at 0-10 min, 16-22% B at 10-15 min, 22-25% B at 15-25 min, 25-34% B at 25-30min, 34-42% B at 30-50 min, v/v and feces: 16-16% B at 0-10 min, 16- 22% B at 10-15 min, 22-25% B at 15-25 min, 25-30% B at 25-30min, 30-35% B at 30-40 min, 35-45% B at 40-55 min, v/v). The flow rate was 1.0 mL/min and column temperature was maintained at 30°C. The detector was set at 278 nm.
Preparation of Radix scutellariae and Scutellariae- Paeoniae couple extracts
Radix scutellariae and Radix Paeoniae Alba were mixed in the ratio of 1.5: 1 g/g and the total weight was 200 g. The mixture was decocted twice by refluxing with 70% ethanol (1: 10 and then 1: 8 w/v) for 1 h, and the solution obtained was concentrated to give an extract of 60.5 g. Radix scutellariae (200 g) was treated as above to provide an extract of 43.3 g. The dried powder was stored at 4 °C before use. For preparation of the dispensing dose, the concentrations of baicalin, wogonoside, baicalein and wogonin in the extracts and pure baicalin were quantitatively determined. The powder extract (0.10 g) was homogenized by ultrasound with 100 mL 60% methanol for 30 min. The solution was filtered through 0.45 μm organic membrane before HPLC analysis. The contents of baicalin (BG), baicalein (B), wogonoside (WG), and wogonin (W) were found to be 17.3, 7.6, 2.4 and 1.7% respectively in Scutellariae-Paeoniae couple extract and 43.6, 19.1, 6.2 and 4.5% respectively in Radix scutellariae extract. The content of baicalin was 95.5 % as pure baicalin.
Animals and Animal Model
Sixty male Sprague-Dawley rats, weighing 250 – 280 g, were obtained from the Experimental Animal Department of Dalian University (Dalian, China). Animal welfare and experimental procedures strictly followed the Guide for the Care and Use of Laboratory Animals (US National Research Council, 1996) and the related ethics regulations of Liaoning University of TCM Study protocols were approved by the Institutional Animal Ethics Committee. Rats were housed in an air-conditioned animal quarter at a temperature of 22 ± 2 °C and a relative humidity of 50 ± 2%. All animals received food and water ad libitum. The animals were acclimatized to the facilities for five days, and then fasted with free access to water for 24 h prior to each experiment.
UC was induced in rats according to the model and method described by Oz HS et al [23]. Rats were lightly anesthetized with 10% chloral hydrate solution, and then a medical-grade polyurethane cannula for enteral feeding (external diameter 2 mm) was inserted into the anus and the tip was advanced to 8 cm proximal to the anal verge. Trinitro-benzene-sulfonic acid (TNBS) dissolved in 100% ethanol was instilled into the colon through the cannula (at a dosage of 80 mg/kg). Following the instillation of hapten, the rats were maintained in a head-down position for an additional 30 s to prevent leakage of the intra colonic instillation. The rats were then returned to their cages. They were checked daily for behavior, body weight, and stool consistency. The animals showed reduction in activities, apathy and occult blood was found in their stool; which proved a successful replication of the UC model since these are the criteria used for evaluating UC rats.
Urine and Feces Sample Collection
Thirty UC rats were divided into three subgroups: the pure baicalin group, Radix scutellariae extract group and Scutellariae-Paeoniae combination extract group. The control rats were divided in the same way. Each group was respectively administrated an oral dose of 0.21 g/kg baicalin in the form of pure compound or coadministrated as mixture, 0.46 g/kg Radix scutellariae extract and 1.16 g/kg Scutellariae-Paeoniae couple extract. All of the compounds were suspended in water and homogenized using ultrasonic technology just prior to dosing. Immediately after the administration of the above compounds, animals were placed individually in glass metabolism chambers (Fuan Inc., Shanghai, China) designed for urine and feces collection. Urine and normal rat feces were collected at 12 – hour intervals throughout the experiment. As it is inconvenient to collect bloody stool of UC rats, we didn’t collect the feces of UC rats. Feces and urine samples were stored at −40°C until analysis of the four flavones.
Biosample preparation
An aliquot of 50 μL arctigenin (32.0 μM in methanol) and 50 μL HCl solution (0.1 mM) were added into 100 μL urine or 0.05 g feces, and then spiked with acetonitrile 1 mL by vortex mixing for 5 min. The mixture was centrifuged at 10000 rpm for 15 min. An aliquot of 50 μL of the supernatant was injected into the HPLC system.
Assay validation
Known amounts of baicalin, baicalein wogonoside, wogonin and IS were added into 100 μL of blank urine and feces to prepare the following series of standards. The calibration curves of urine and feces showed good linearity in the range of 0.384 ~ 195.5 and 0.096 ~ 49.3 μM for baicalin; 0.241 ~ 123.6 and 0.137 ~ 70.2 μM for wogonoside; 0.070 ~ 36.0 and 0.140 ~ 72.0 μM for baicalein; 0.065 ~ 33.4 and 0.198 ~ 101.2 μM for wogonin. The extraction recoveries and matrix effects at three quality control (QC) concentrations were assayed in sets of six replicates. The CVs of the recoveries were less than 20% at low concentration, and less than 15% at medium and high concentrations. The recoveries were all more than 70%. The means ± SD recovery for IS was 94.1 ± 4.8%. Accuracy and precision of the method were determined by intra-day and inter-day data by comparing the mean of five replicates of each QC sample. Both coefficient of variation (CV) for precision evaluation and relative error (RE) for accuracy evaluation were less than ±14%. The stabilities of baicalin, wogonoside, baicalein and wogonin were determined by analyzing QC samples at three concentrations exposed to encounter during sample storage. The corresponding relative errors were less than ±6% for samples at three concentrations for each reference standard, respectively. Results of the stability test showed that under the experimental conditions the samples were stable throughout the testing process.
Data analysis
The concentrations of baicalin, baicalein, wogonoside and wogonin in urine or feces were multiplied by the respective urinary volume or feces weight collected in each time interval to obtain the total amount excreted in the sampling time. All the results were expressed as means ± standard deviation (SD). The Microsoft Excel was used to calculate the cumulative excretion amounts. Statistical analysis was performed using SPSS software package (version 15.0). Differences between means were considered significant if P ≤ 0.05.
Results
HPLC assay
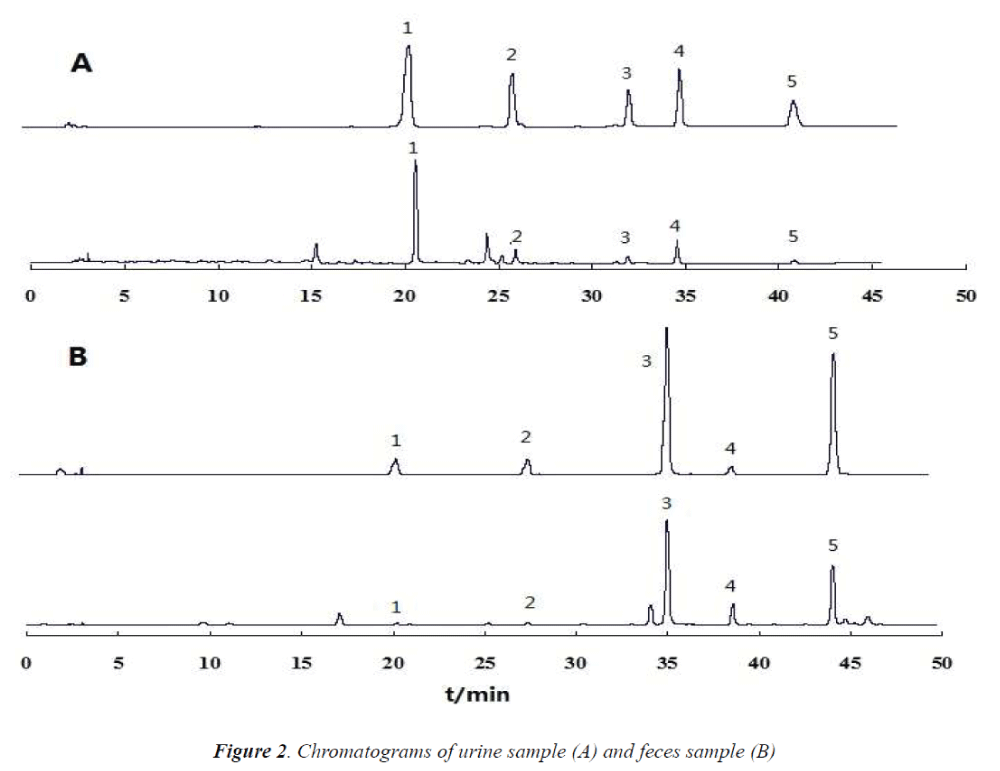
The selectivity of the method was evaluated by analyzing blank urine and feces samples prior to administration. The chromatograms were free of interfering peaks at the retention times of IS and baicalin, wogonoside, baicalein and wogonin. Fig.2 (A) the representative chromatograms of blank urine and feces samples spiked with baicalin, wogonoside, baicalein, wogonin and IS (A), urine andS feces sample (12 h) after oral administration of Scutellariae- Paeoniae couple extract (B). Under the established chromatographic condition, no interfering peaks were observed at the elution times of baicalin, wogonoside, bai calein, wogonin and the internal standard. Feces samples show the same result Fig. 2 (B).
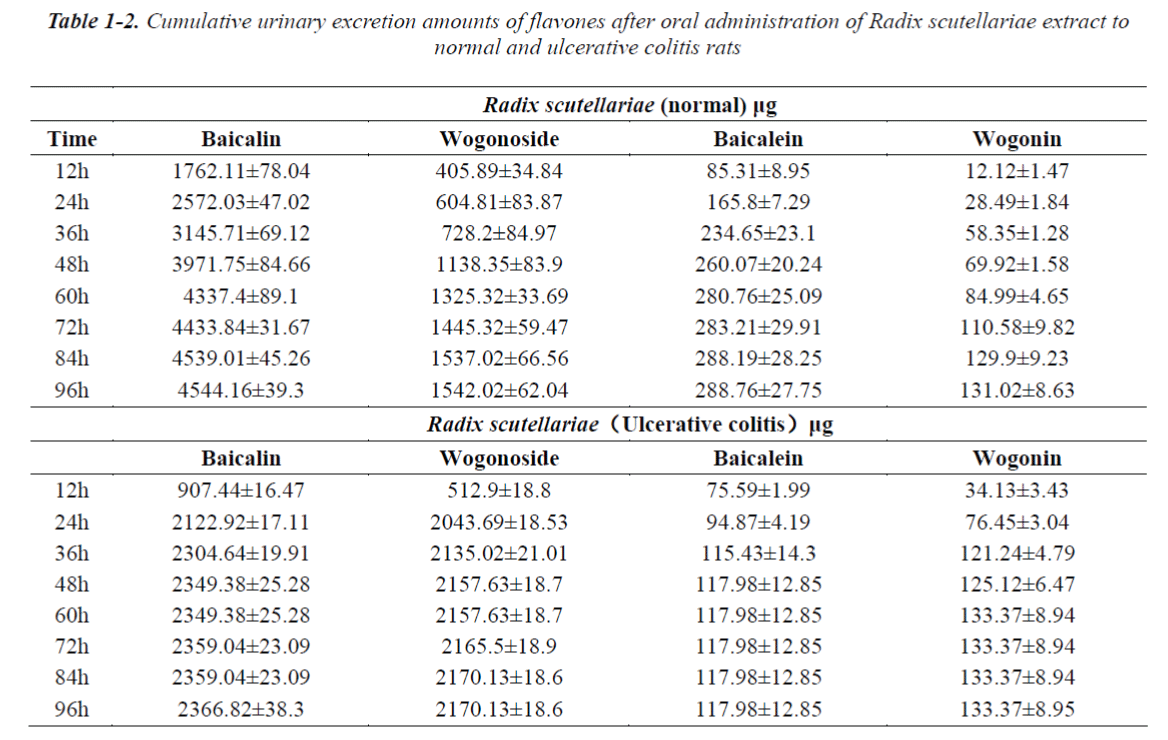
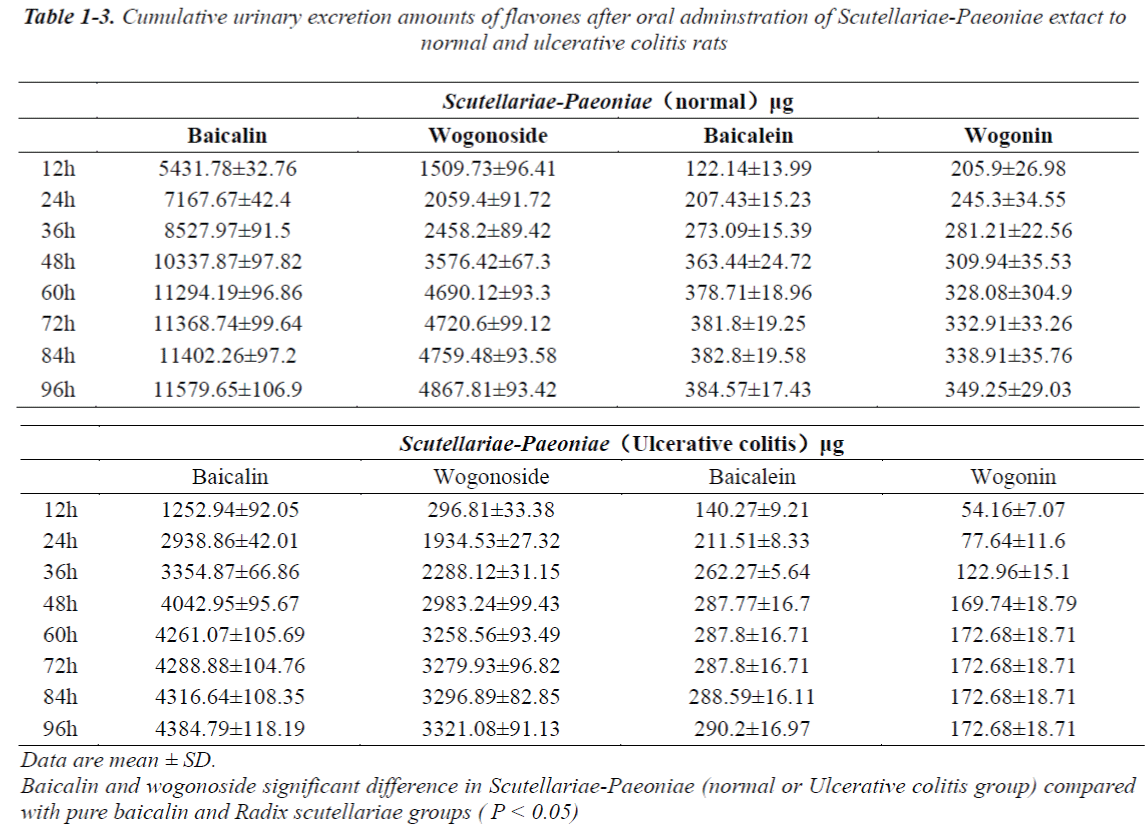
Excretion profiles of the four flavonides in urine and feces after oral administration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae extracts to normal and UC rats.
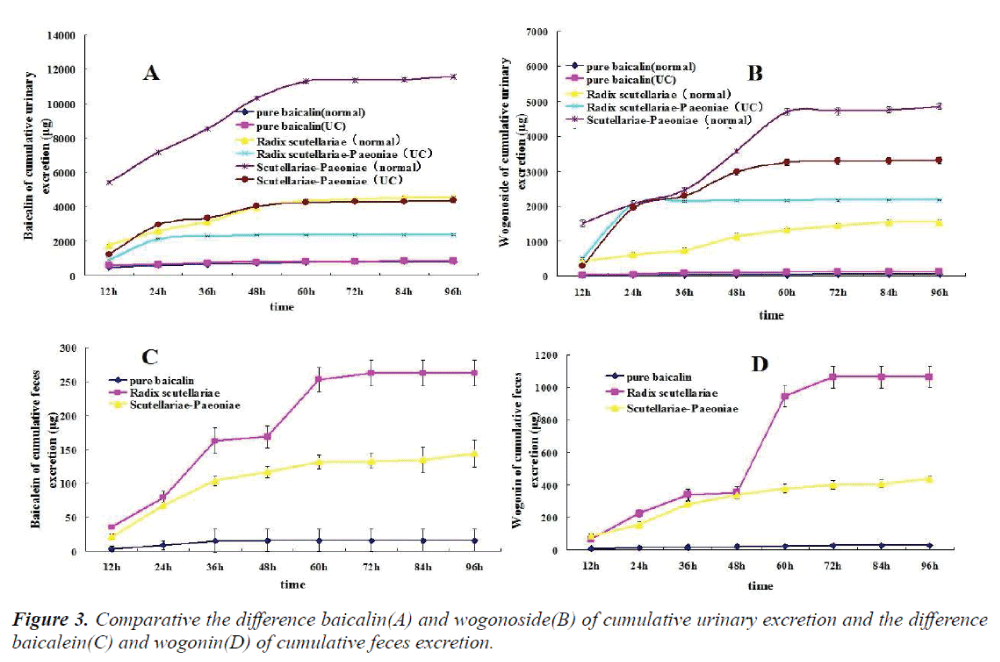
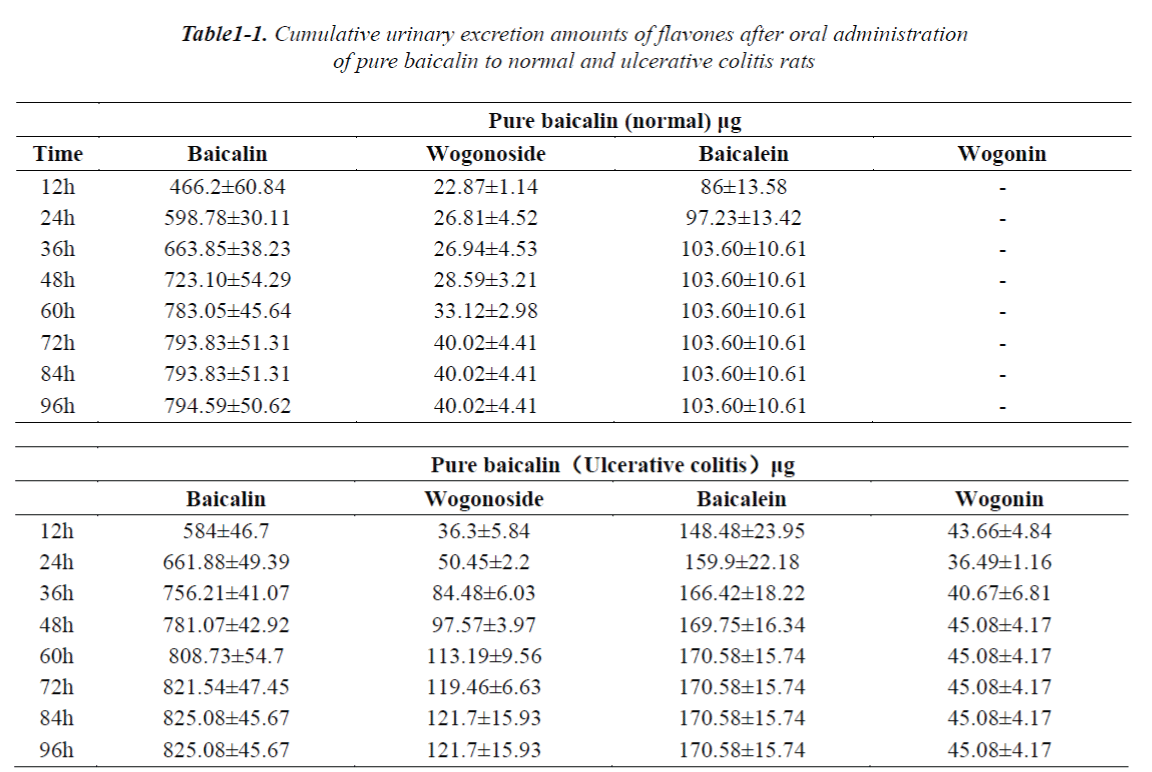
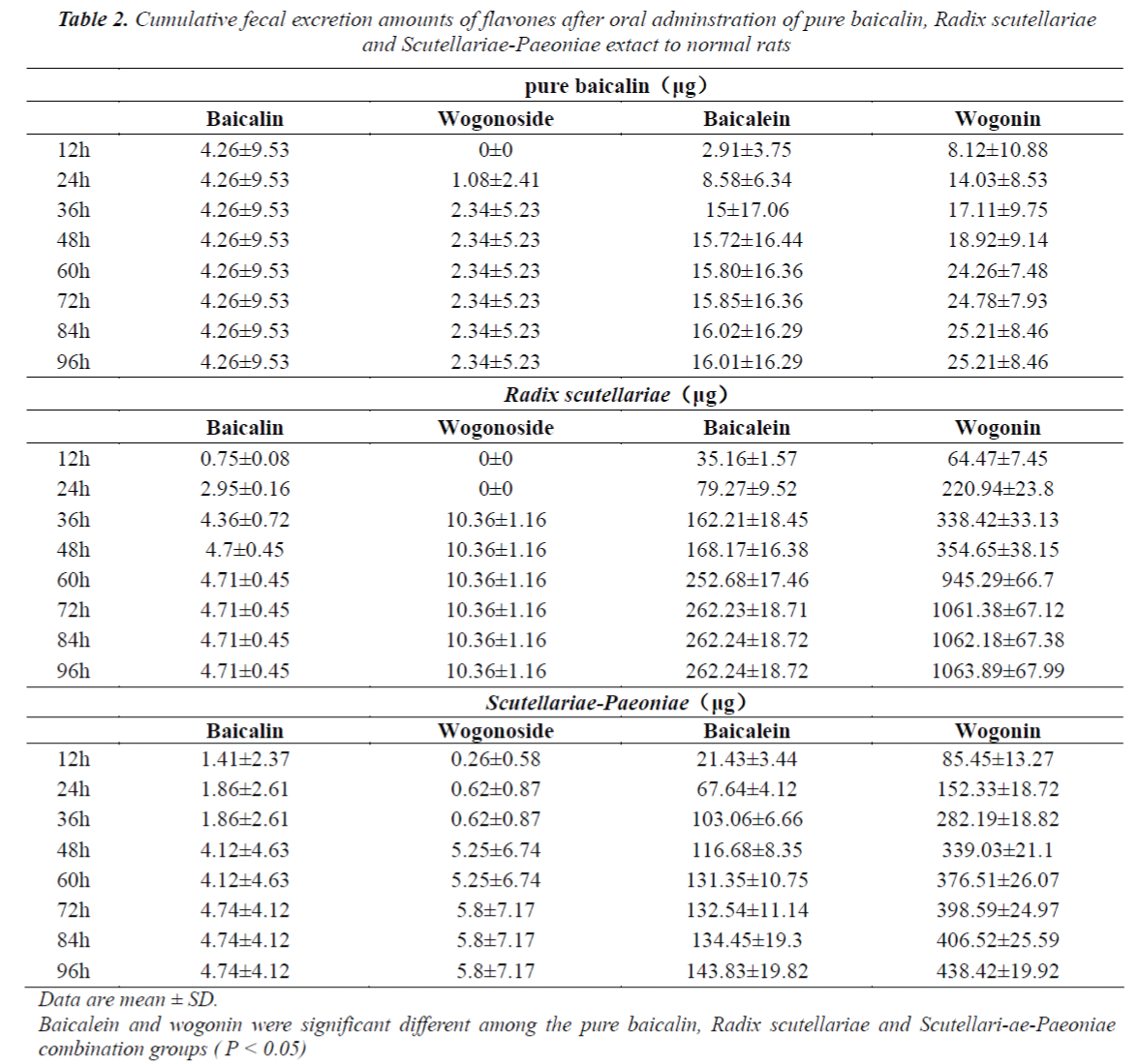
The cumulative urinary excretion amounts of flavones after oral administration of pure baicalin, Radix scutellariae or Scutellariae-Paeoniae extracts to normal and UC rats are listed in Table 1(1-3). Not only baicalin and wogonoside but also baicalein and wogonin were detected in the urine and/or feces after oral administration of pure baicalin to rats. Comparative differences in cumulative urinary excretion of baicalin and wogonoside after oral adminstration of pure baicalin, Radix Scutellariae and Scutellariae-Paeoniae to nomal and UC rats are depicted in Fig.3 (A,B). The cumulative fecal excretion of flavones after oral adminstration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae extact to rats are listed in Table 2. Comparative differences of cumulative feces excretion of baicalein after oral adminstration of pure baicalin, Radix Scutellariae and Scutellariae-Paeoniae to rats are depicted in Fig. 3 - (C,D)
Discussion
Quantitation of the commercial powder of Scutellariae Radix indicated that baicalin, baicalein wogonoside and wogonin were the major flavone constituents. These flavones have been shown to possess antilipoperoxidant, antiplatelet, and anti-inflammatory activities. They also demonstrated not only cytostatic but also cytotoxic effects on various human tumor cell lines in vitro and suppress tumor growth in vivo. Biological activities of baicalin and wogonoside depended on their conversion to the aglycone form by b-glucuronidase from normal colonic flora and their subsequent absorption [25]. Table 1-1 and 2-1 shows the presence of baicalin and wogonoside as well as baicalein and wogonin in the urine and feces of rats after oral administration of pure baicalin. We had tentatively concluded that the absorbed baicalin could be methylated to wogonoside in vivo and then baicalin and wogonoside are partly converted to baicalein and wogonin by glucuronidase (GUS) and distributed in the rat tissues [26,27]. Li [28] reported that stereoselective transformation by microfloral metabolism in the intestinal tract might have occurred before absorption, showing that the biotransformations from the corresponding flavones, wogonin and baicalein, were stereoselective with a preference. In this report, our results appeared to be consistent with previous reports.
After oral administration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae couple extracts to normal and UC rats, baicalin, wogonoside, baicalein or wogonin were all detected in rat urine. Excreted levels of baicalin and wogonoside were high while levels of baicalein or wogonin were comparatively lower in the urine. Glycoside is also excreted in urine. Differences in the excretion profiles of baicalin and wogonoside in urine were reflected in the excretion speed and cumulative urinary excretion amounts. These compounds were excreted at different rates. The excretion rate of baicalin and wogonoside in the pure baicalin group was faster than those in Radix scutellariae and Scutellariae- Paeoniae couple groups. That is, the cumulative amounts of the four flavones in rat urine reached plateau value at 60 h, 72 h, 84 h, respectively. Baicalin and wogonoside were retained longer in the body. Compared with that in the single baicalin and single herb decoction group, we found that either in UC or in normal rats, Scutellariae-Paeoniae couple group showed more cumulative excretion amounts. The cumulative amounts of these flavones in rat urine among these groups were significantly different (P < 0.05). The higher percentage of cumulative urinary excretion may be attributed to their higher absorption. A significant drug-drug interaction may occur and improve the absorption of baicalin and wogonoside when Radix Paeoniae alba is administered in combination with Radix scutellariae [29]. Radix Paeoniae alba, used as messenger herb and hematinic herb, could increase the absorption of baicalin and wogonoside.
It was also found that the normal groups showed significant differences of urinary excretion compared with UC groups. A tendency of lower urinary excretion was observed in UC rats, regardless of oral administration of pure baicalin, Radix scutellariae or Scutellariae- Paeoniae couple extracts. Physiological phenomenon may be the cause of a decrease in the excretion of the four flavones. We have reported that in UC, orally administered baicalin and wogonoside are absorbed more completely leading to higher plasma baicalin and wogonoside compared to normal rats (personal communication). So, when UC rats were admistrated pure baicalin, Radix scutellariae or Scutellariae-Paeoniae couple extracts, the bioavailability of the flavones were enhanced in contrast to normal rats.
Baicalin, wogonoside, baicalein and wogonin all were excreted in the rat feces. But excretion of baicalein and wogonin was primarily in the feces. Cumulative fecal excretion amounts in rats were enhanced by Radix scutellariae (RS) extract or Scutellariae-Paeoniae couple extract. After oral administration of pure baicalin, Radix scutellariae and Scutellariae-Paeoniae extracts as combination to rats, the cumulative amounts of baicalin, baicalein, wogonoside and wogonin in rat feces reached plateau value at 60 h, 60 h, and 84 h respectively, and the cumulative amounts of these flavones in rat feces also differed significantly (P < 0.05). Cumulative fecal excretion in pure baicalin group was the least. The results indicate that the amount of flavones excreted in feces may be directly proportional to the area under curve (AUC)of plasma, and compatibility of medicines could result in the differences of excretion.
Conclusion
In summary, we report for the first time the urinary and fecal excretion parameters of the four flavones after oral admistration of pure baicalin, Radix scutellariae or Scutellariae-Paeoniae extracts to rats; and suggest that urinary and fecal excretion of the four flavones could be affected by herb-drug interaction in vivo, and ulcerative colitis could influence the potential excretion of the four flavones. From the results we have derived a hypothesis that Radix scutellariae and Radix paeoniae alba in combination seemed suitable for curing ulcerative colitis disease. The results of the present study highlight that drug-drug interactions, herb-drug interactions and herbherb interactions have always existed and affected the excretion of herbal ingredients. The huge nuber of active ingredients in TCM have made them suitable for multitarget actions, and this remedy might be a reasonable prescription [30].
References
- Young HC, Yoon Gyoon K, Young WC. Herb-Drug interactions: focus on metabolic enzymes and trans- porters. Arch Pharm Res 2011; 34: 1843-1863.
- Lü JM, Yao Q, Chen C. Ginseng compounds: an update on their molecular mechanisms and medical applica- tions. Curr Vasc Pharmacol 2009; 7: 293-302.
- Ross SM, Milk SM. An ancient botanical medicine for modern times. Holist. Nurs Pract 2008; 22: 299-300.
- Patel MA, Patel PK, Patel MB. Effects of ethanol ex- tract of Ficus bengalensis (bark) on inflammatory bowel disease. Indian J Pharmacol 2010; 42: 214-218.
- Liu L, Deng YX, Liang Y, Pang XY, Liu XD, Liu YW, Yang JS, Xie L, Wang GJ. Increased oral AUC of bai- calin in streptozotocin-induced diabetic rats due to the Increased activity of intestinal β-glucuronidase. Planta Med 2010; 76: 70-75.
- de Faria FM, Luiz-Ferreira A, Socca EA, de Almeida AC, Dunder RJ, Manzo LP, da Silva MA, Vilegas W, Rozza AL, Pellizzon CH, Dos Santos LC, Souza Brito AR. Effects of Rhizophora mangle on experimental co- litis induced by TNBS in rats. Evid Based Comple- ment Alternat Med 2012; 2012:753971.
- Zhu Z, Zhao L, Liu XF, et al. Comparative pharma-cokinetics of baicalin and wogonoside by liquid chro- matography–mass spectrometry after oral administra- tion of XiaochaihuTang and Radix scutellariae extract to rats. J Chromatogr B Analyt Technol Biomed Life Sci 2010; 878: 2184-2190.
- Mondal SK, Mondal NB, Banerjee S, Mazumder UK. Determination of drug-like Properties of a novel antileishmanial compound: In vitro absorption, distri- bution, metabolism, and excretion studies. Indian J Pharmacol 2009; 41: 176-781.
- Deng YX, Shi QZ, Chen B, Zhang XZ, Liu SZ, Qiu XM. Comparative pharmacokinetics of baicalin in nor- mal and the type 2 diabetic rats after oral administra- tion of the Radix scutellariae extract. Fitoterapia 2012; 83:1435-1442.
- Lu T, Song J, Huang F, Deng YX, Xie L, Wang G, Liu X. Comparative pharmacokinetics of baicalin after oral administration of pure baicalin, Radix scutellariae ex- tract and Huang-Lian-Jie-Du-Tang to rats. J Ethno- pharmacol 2007; 110: 412-418.
- Kim YH, Jeong DW, Kim YC, Sohn DH, Park ES, Lee HS. Pharmacokinetics of baicalein, baicalin and wogonin after oral administration of a standardized ex- tract of Scutellaria baicalensis, PF-2405 in rats. Arch Pharm Res 2007; 30: 260-265.
- Wang Y, Yao Y, An R, You L, Wang X. Simultaneous determination of puerarin, daidzein, baicalin, wogo- noside and liquiritin of GegenQinlian decoction in rat plasma by ultra-performance liquid chromatography– mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2009; 877: 1820-1826.
- of baicalein, baicalin, oroxylin A and wogonin in rat plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 844: 261-267.
- Akao T, Hanada M, Sakashita Y, Sato K, Morita M, Imanaka T. Efflux of baicalin, a flavone glucuronide of Scutellariae radix, on Caco-2 cells through multidrug resistance-associated protein 2. J Pharm Pharmacol 2007; 59: 87-93.
- Liu TM, Jiang XH. Investigation of the absorption mechanisms of baicalin and baicalein in rats. J Pharm Sci 2006; 95: 1326-1333.
- Lai MY, Hsiu SL, Tsai SY, Hou YC, Chao PD. Com- parison of metabolic pharmacokinetics of baicalin and baicalein in rats. J Pharm Pharmacol 2003; 55: 205-209.
- Zhang L, Lin G, Chang Q, Zuo Z. Role of intestinal first-pass metabolism of baicalein in its absorption process. Pharm Res 2005; 22: 1050-1058.
- Hu Z, Yang X, Ho PC, Chan SY, Heng PW, Chan E, Duan W, Koh HL, Zhou Sm. Herb–drug interactions: a literature review. Drugs 2005; 65: 1239-1282.
- Zeng MF, Pan LM, Zhu HX, Zhang QC, Guo LW. Comparative pharmacokinetics of baicalin in plasma after oral administration of Huang-Lian-Jie-Du-Tang or pure baicalin in MCAO and sham-op-erated rats. Fi- toterapia 2010; 81: 490-496.
- Feng NP, Di B, Liu WY Comparison of the metabolism of baicalin in rats orally administered with Radix scutellariae extract and Shuang-Huang-Lian extract. Chem Pharm Bull 2005; 3: 978-983.
- Lee IA, Kim EJ, Kim DH. Inhibitory effect of β- Sitosterol on TNBS-Induced colitis in mice. Planta Med 2012; 78: 896-898.
- Liu Y, Xiang J, Liu M, Wang S, Lee RJ, Ding H. Pro- tective effects of glycyrrhizic acid by rectal treatment on a TNBS-induced rat colitis model. J Pharm Pharma- col 2011; 63: 439-446.
- Oz HS, Zhong J, de Villiers WJ. Osteopontin Ablation Attenuates Progression of Colitis in TNBS Model. Di-ges Dis Sci 2012; 57: 1554-1561.
- Liu W,Li F,Zhuang L,et al. Comparative the tis-sue distributions of flavonoids after oral adminis-tration of pure baicalin, Radix Scutellariae and Scutellariae- Paeoniae couple extracts to rats. J Med Plants Res 2011; 31: 6907-6915.
- Feng J, Xu W, Tao X, Wei H, Cai F, Jiang B, Chen W. Simultaneous determination of baicalin, baicalein, wogonin, berberine, palmatine and jatrorrhizine in rat plasma by liquid chromatography-tandem mass spec- trometry and application in pharmacokinetic studies af- ter oral administration of traditional Chinese medicinal preparations containing Scutellaria-Coptis herb couple. J Pharm Biomed Anal 2010; 53: 591-598.
- Shi R, Zhou H, Liu ZM, et al. Influence of Coptis Chinensis on pharmacokinetics of flavonoids after oral administration of Radix Scutellariae in rats. Biopharm Drug Dispos 2009; 30: 398-410.
- Prigol M, Brüning CA, Martini F, Nogueira CW. Com- parative excretion and tissue distribution of selenium in mice and rats following treatment with diphenyl dise- lenide. Biol Trace Elem Res 2012; 150: 272-277.
- Li C, Homma M, Ohkura N, Oka K. Stereochemistry and putative origins of flavanones found in post- administration urine of the traditional Chinese remedies shosaiko-to and daisaiko-to. Chem Pharm Bull (Tokyo) 1998; 5: 807-811.
- Li CR, Lin G, Zuo Z. Pharmacological effects and pharmacokinetics properties of Radix Scutellariae and its bioactive flavones. Biopharm Drug Dispos 2011; 32: 427-445.
- Srinivas NR. Baicalin, an emerging multi-therapeutic agent: pharmacodynamics, pharmacok-inetics, and con- siderations from drug development perspectives. Xeno- biotica 2010; 40: 357-367.