Research Article - Journal of Aging and Geriatric Psychiatry (2017) Journal of Aging and Geriatric Psychiatry (Special Issue 3-2017)
Mortality, inappropriate medication and length of hospital stay in older hip fracture patients: A general population-based cohort study
Annika Kragh Ekstam1,2*, Isam Atroshi3,4, Sölve Elmståhl11Department of Clinical Sciences, Geriatric Medicine, Lund University, Skåne University Hospital, Malmö, Sweden
2Department of Medicine, Hässleholm Hospital, Hässleholm, Sweden
3Department of Orthopedics, Hässleholm and Kristianstad Hospitals, Hässleholm, Sweden
4Department of Clinical Sciences Lund, Lund University, Lund, Sweden
- *Corresponding Author:
- Annika Kragh Ekstam
Department of Clinical Sciences
Lund University
Skåne University Hospital
Malmö
Sweden
Tel: +46443091272
E-mail: annika.kraghekstam@skane.se
Accepted Date: October 13, 2017
Citation: Ekstam AK, Atroshi I, Elmståhl S. Mortality, inappropriate medication and length of hospital stay in older hip fracture patients: A general population-based cohort study. J Aging Geriatr Psychiatry. 2017;1(3):8-15
Abstract
Purpose: To assess if potentially inappropriate medication (PIM) or length of hospital stay (LOS)
Keywords
Hip fracture, Older patients, Inappropriate medication, Length of in-hospital stay, Mortality
Introduction
Due to a growing older population in many countries an increasing number of hip fracture patients have become a major health concern [1-4]. The annual incidence of hip fractures worldwide has been estimated to increase by three to four times and to exceed 6 million fractures by 2050 [5,6]. Interventions aimed at reducing preventable falls and fractures, as well as increasing drug safety in the care of hip fracture patients are urgently needed.
Through the years a number of interventions has been implemented to improve the care of hip fracture patients and these have in many aspects led to better in-hospital management and communal care [7-11]. But so far little has been achieved to improve drug safety in hip fracture patients. As previously reported, few hip fracture patients are prescribed antiosteoporotic drugs and even fewer have their medication adjusted to reduce the risk of new falls, fractures and other adverse outcomes [12]. When studying the use of fall-risk increasing drugs (FRID) in hip fracture patients it was found that the prescribing of FRID was frequent before the fracture and increased by 30 percentage points in the six months following the fracture and could be related to increased mortality [12,13].
Older people are more vulnerable to adverse effects from drugs, primarily owing to the functional decline of inner organs due to the aging process [14,15]. These changes are known to increase the susceptibility to drugs and to cause adverse reactions such as nausea, sedation, dizziness, low blood pressure, falls, confusion, and kidney failure [16,17].
Contributing causes to unwanted drug effects in older patients are the need for multiple medications caused by multimorbidity, and drug-drug interactions. In addition to the risks caused by polypharmacy, defined as ≥ 5 drugs, the use of potentially inappropriate medication (PIM) can lead to adverse events. Treatment with PIM has been shown to cause increased adverse reactions and higher mortality [18-20]. One important way of improving drug therapy in older patients is to reduce the prescribing of PIM and to identify its’ potential consequences. In several countries lists over PIM are employed to identify and limit the prescribing of drugs that can cause serious adverse effects in older people. The Beer´s explicit criteria, developed in the USA in 1991, are often used for this purpose [21,22]. As drug-therapy differ between countries there is a need for national guidelines on PIM. The Swedish National Board of Health and Welfare has identified drugs as PIM which are not included in the Beers’ list [23].
Hip fractures can have serious consequences in older people with more than one out of four patients dying within a year after the fracture [1,24-26]. The high mortality rate is shown to be not only related to the acute trauma and surgery, but also to the subsequent increase in cardiovascular- and pulmonary diseases, infections, and depression. Persistent pain following a hip fracture has also a negative effect on the patients’ quality of life and level of activity [27-29].
The aim of this study on older hip fracture patients is to explore avoidable risk factors to short term mortality, namely potentially inappropriate medication and the length of in-hospital stay.
Method
Study design and cohort
This is a cohort study conducted in a general population in a county of southern Sweden (1.3 million inhabitants, 13% of the Swedish population). All county residents aged 60 years or older, who were registered in the national In-patient as having a hip fracture diagnosis, from January 1 through December 31 in 2006, were included. Patients with the diagnostic codes S72.00, S72.10, S72.11, S72.20 and S72.21, according to the International Classification of Diseases, tenth revision (ICD10th), were included and none excluded.
Data collection
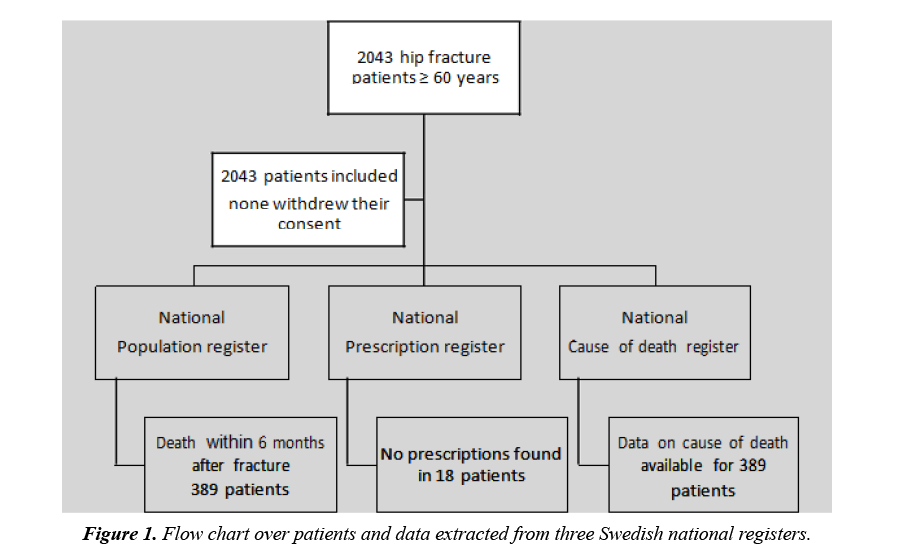
Data was retrieved from three national registers. Available data included the participants’ age, sex, date of admission and discharge, type of fracture, in-hospital stay, time and cause of death six months after admission (Figure 1). Because of major differences between the orthopaedic departments’ regarding the registration of diagnoses other than the hip fracture, the data on comorbidities was not deemed reliable. Data on hip fracture diagnosis, type of surgery, time of death, cause of death and prescribed drugs, were obtained from Swedish national statistics databases and based on the participants’ unique civic number. Drugs dispensed six months before to six months after the fracture (or until death) were retrieved from the National Prescription Register. The register holds prescriptions from all care settings, and dispensed drugs are registered in accordance with the law. A drug was included in the analyses when it was dispensed twice or more within the year to increase the possibility that the drugs were consumed by the patients. Comparing to unexposed patients we analysed patients’ exposure to PIM, polypharmacy, and opioids, as well as differences in sex, age, type of fracture, mortality, and cause of death.
PIM was identified from Beers’ list of explicit criteria (2015) as well as lists from Swedish health authorities (also included propiomazine, tramadol and dextropropoxyphene) [21,23,30]. Analgesic drugs were analysed separately. Patients’ exposure to PIM were divided into six groups; none, any, 1, 2, 3 to 4, and 5 or more. PIM was divided into five separate therapeutic groups; analgesic, psychotropic, anticholinergic, cardiovascular and various. The group labelled as “various” PIM, included the following drugs; antiparkinson, antispasmodic, noninflammatory steroids, antithrombotic, skeletal musclerelaxants, gastrointestinal, endocrine and anti-infective.
Statistical analysis
Baseline differences were analysed using the t-test for continuous variables and the χ2-test for categorical variables. Logistic regression analysis was carried out to determine whether use of PIM was associated with differences in mortality, cause of death or length of in-hospital stay. Use of PIM was adjusted for differences in age, sex, and polypharmacy, with 30-, 90- and 180-day mortality as the dependent variables. Due to the lack of reliable information on comorbidity, polypharmacy was used as a proxy. As a measure of association between exposure to PIM and death we calculated odds ratios (OR) with a 95% confidence interval (CI) to estimate the precision. All analyses were performed using SPSS 22.0 (Statistical Package for the Social Sciences) and were two-sided with a p-value <0.05 indicating statistical significance.
Results
Out of the 2043 patients’ aged ≥ 60 years who were admitted to the five trauma-care hospitals for hip fracture treatment, 27% were male (mean age 80.8 years) and 73% were female (mean age 83.5 years) (Table 1). Male mortality was significantly higher than female at 30-day (p=0.004), 90-day (p=0.020) and 180- day (p=0.007) after the fracture and old age was also correlated (p<0.001) to death. Significant differences were found between the two studied age groups (60-79 and ≥ 80 years) with regard to type of fracture, mortality, exposure to any PIM, polypharmacy, and cardiovascular drugs.
Table 1. Clinical characteristics of the study population classified by sex and age.
| Characteristics | All | Male | Female | Age 60-79 years | Age ≥ 80 years | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2043 N (%) | 541 (26.5%) |
1502 (73.5%) | 594 (29.1%) |
1449 (70.9%) | ||||||||
| N (%) | N (%) | P value | N (%) | N (%) | P value | |||||||
| Age, mean ± SD | 82.8 ±8.0 | 80.8 ±8.2 | 83.5 ±7.8 | <0.001 | ||||||||
| age 60-79 years | 594 (29.1) |
201 (37.2) |
393 (26.2) |
|||||||||
| age ≥ 80 years | 1,449 (70.9) | 340 (62.8) |
1,109 (73.8) | <0.001 | ||||||||
| Type of fracture | ||||||||||||
| cervical (S7200) | 1062 (52.0) | 272 (50.3) |
790 (52.6) |
0.355 | 357 (60.1) |
705 (48.7) | <0.001 | |||||
| pertrochanteric (S7210) | 839 (41.1) |
231 (42.7) |
608 (40.5) |
0.368 | 193 (32.5) |
646 (44.5) | <0.001 | |||||
| subtrochanteric (S7220)† | 142 (6.9) |
38 (7.0) |
104 (6.9) |
0.896 | 44 (7.4) |
98 (6.8) | 0.564 | |||||
| Mortality | ||||||||||||
| 30 days | 173 (8.5) |
62 (11.5) |
111 (7.4) |
0.004 | 26 (4.4) |
147 (10.1) | <0.001 | |||||
| 90 days | 304 (14.9) |
97 (17.9) |
207 (13.8) |
0.020 | 44 (7.4) |
260 (17.9) | <0.001 | |||||
| 180 days | 389 (19.0) |
124 (22.9) |
265 (17.6) |
0.007 | 58 (9.8) |
331 (22.8) | <0.001 | |||||
| LOS days mean ± SD | 9.52 ±5.7 | 9.87 ±6.1 | 9.40 ±5.5 | 0.169 | 9.22 ±5.2 | 9.65 ±5.9 | 0.621 | |||||
| Median | 9 | 9 | 9 | 9 | 9 | |||||||
| 9 days or less | 1,101 (54) | 290 (53.6) |
811 (54.0) |
339 (57.1) |
762 (52.6) | |||||||
| 10 days or more | 942 (46) |
251 (46.4) |
691 (46.0) |
0.876 | 255 (42.9) |
687 (47.4) | 0.065 | |||||
| PIM analgesic | 1233 (60.4) | 307 (56.7) |
926 (61.7) |
0.046 | 377 (65.5) |
856 (58.4) | 0.003 | |||||
| PIM psychotropic | 601 (29.4) | 141 (26.1) |
460 (30.6) |
0.046 | 147 (25.5) |
454 (30.9) | 0.015 | |||||
| PIM various | 408 (20.0) | 98 (18.1) |
310 (20.6) |
0.208 | 129 (22.4) |
279 (19.0) | 0.086 | |||||
| PIM anticholinergic | 276 (13.5) | 72 (13.3) |
204 (13.6) |
0.873 | 84 (14.6) |
192 (13.1) | 0.374 | |||||
| PIM cardiovascular | 140 (6.9) | 66 (12.2) |
74 (4.9) | <0.001 | 48 (9.3) |
80 (6.3) | 0.097 | |||||
| Number of PIM, any | 1666 (81.5) | 428 (79.1) |
1238 (82.4) | 0.089 | 505 (85.0) |
1,161 (80.1) | 0.010 | |||||
| 0 | 377 (18.5) | 113 (30.0) |
264 (70.0) |
0.089 | 89 (23.6) |
288 (76.4) | 0.010 | |||||
| 1 | 629 (30.8) | 160 (29.6) |
469 (31.2) |
0.476 | 199 (33.5) |
430 (29.7) | 0.089 | |||||
| 2 | 482 (23.6) | 133 (24.6) |
349 (23.2) |
0.526 | 138 (23.2) |
344 (23.7) | 0.806 | |||||
| 3-4 | 447 (21.9) | 107 (19.8) |
340 (22.6) |
0.168 | 129 (21.7) |
318 (29.9) | 0.909 | |||||
| ≥5 | 108 (5.3) | 28 (5.2) |
80 (5.3) | 0.893 | 39 (6.6) |
69 (4.8) | 0.098 | |||||
| Polypharmacy, ≥ 5 drugs | 1569 (76.8) | 389 (71.9) |
1180 (78.6) | 0.002 | 418 (70.4) |
1151 (79.4) | <0.001 | |||||
| Cardiovascular drugs | 1505 (73.7) | 394 (72.8) |
1111 (74.0) | 0.606 | 383 (64.5) |
1122 (77.4) | 0.001 | |||||
| Opioids (not PIM) | 645 (31.6) |
169 (31.2) |
476 (31.7) |
0.846 | 187 (31.5) |
458 (31.6) | 0.955 | |||||
Note: †One patient coded as S72.21, open subtrochanteric proximal femur fracture. LOS: length of in-hospital stay; PIM: potentially inappropriate medication; SD: standard deviation
The most frequently used PIM were tramadole (790 patients, 39%), dextropropoxyphene (413 patients, 20%), drugs with strong anticholinergic effects (276 patients, 13.5%), non-steroid anti-inflammatory drugs (249 patients, 12%) and long-acting benzodiazepines (207 patients, 10%). Out of the 1666 (81.5%) patients treated with any PIM, 1,161 (80%) were older patients (≥ 80 years). The number of PIM the patients were exposed to, did not significantly affect their mortality. The 377 (18.5%) patients not exposed to PIM included a large number of older (≥ 80 years) individuals (p=0.010). Women were more often treated with ≥ 5 drugs (p=0.002), analgesic PIM (p=0.046), and psychotropic drugs (p=0.046), whereas men were more often exposed to cardiovascular PIM (p<0.001) (Table 2). Polypharmacy was present in 1,569 patients (77%), in most patients treated with PIM (p<0.001), and in older patients (p<0.001).
Table 2. Patients’ characteristics and exposure to potentially inappropriate medication and polypharmacy.
| Exposed to | PIM | PIM | PIM | PIM | PIM | PIM |
|---|---|---|---|---|---|---|
| Any | Analgesic | Psychotropic | Various | Anticholinergic | Cardiovascular | |
| N (%) | 1666 (81.5) | 1233 (60) | 601 (29) | 408 (20) | 276 (13.5) | 140 (7) |
| Male | 428 (79) | 307 (57) | 141 (26) | 98 (18) | 72 (13) | 66 (12) |
| Female | 1238 (82) | 926 (61.5) | 460 (31) | 310 (21) | 204 (14) | 74 (5) |
| p value | 0.089 | 90 | 0.046 | 0.208 | 0.873 | <0.001 |
| 60-79 years | 505 (85) | 405 (68) | 152 (26) | 130 (22) | 87 (15) | 46 (8) |
| ≥ 80 years | 1161 (80) | 828 (57) | 449 (31) | 278 (19) | 189 (13) | 94 (6.5) |
| p value | 0.01 | 0.003 | 0.015 | 0.166 | 0.336 | 0.307 |
| Polypharmacy | 1363 (82) | 980 (62.5) | 543 (35) | 351 (22) | 257 (-16) | 134 (8.5) |
| (≥ 5 drugs) | ||||||
| p value | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
Note: PIM: potentially inappropriate medication
Mortality, PIM, and cause of death
Out of the patients, 389 (19%) died within six months following the fracture. The most frequent cause of death was diseases in the circulatory system (133 patients, 34%) and was significantly more frequent in older patients. Cancer was the primary cause of death in 48 patients (12.5%). Unadjusted analyses on cause of death, 30-, 90-, and 180-day, showed that patients exposed to any PIM (p=0.003, p ≤ 0.001, and p=0.008, respectively) were more likely (64, 68 and 73%) to die from diseases in the circulatory system, compared to unexposed patients.
Analysis on mortality at 30- and 90-day, adjusted for age, sex and polypharmacy, presented a significantly higher mortality in patients exposed to any PIM compared to unexposed patients.
Odds ratio for 30-day mortality was 1.79 (95% CI 1.25- 2.57, p=0.002) and for 90-day mortality 1.57 (95% CI 1.16- 2.12, p=0.003) (Table 3). Patients exposed to analgesic PIM (tramadole and dextropropoxyphene) showed a significantly higher mortality (p<0.001), OR at 30-day 2.59 (95% CI 1.85- 3.63), 90-day 1.94 (95% CI 1.50-2.51), and at 180-day 1.62 (95% CI 1.29-2.05). Analysis on patients exposed to other strong analgesics, not deemed to be inappropriate, (morphine, oxycodone, codeine, fentanyl, ketobemidone) instead had a significantly lower mortality at 180-day with OR 0.72 (95% CI 0.56-0.92) and mortality was not increased at the 30- or 90-day intervals.
Table 3. Six months mortality in 2,043 older hip fracture patients, potentially inappropriate medication, opioids, and LOS, adjusted for age, sex, and polypharmacy.
| Number exposed | Mortality 30 days | Mortality 90 days | Mortality 180 days | |
|---|---|---|---|---|
| Exposed to | N (%) | 173 (8.5%) OR [95% CI] | 304 (14.9%) OR [95% CI] | 389 (19.0%) OR [95% CI] |
| PIM any | 1666 (81) | 1.79 [1.25 – 2.57] | 1.57 [1.16 – 2.12] | 1.29 [0.97 – 1.71] |
| PIM all analgesicss | 1233 (60) | 2.59 [1.85 – 3.63] | 1.94 [1.50 – 2.51] | 1.62 [1.29– 2.05] |
| PIM tramadole | 790 (39) | 2.03 [1.38 – 2.99] | 1.53 [1.15 – 2.02] | 1.32 [1.03 – 1.68] |
| PIM dextropropoxyphene | 413 (20) | 1.49 [0.94 – 2.35] | 1.49 [1.05 – 2.11] | 1.29 [0.95 – 1.75] |
| PIM psychotropic | 601 (29) | 0.91 [0.63 – 1.32] | 0.79 [0.59 – 1.05] | 0.77 [0.59 – 0.99] |
| PIM various drugs | 408 (20) | 0.83 [0.55 – 1.26] | 0.79 [0.58 – 1.09] | 0.7 [0.53 – 0.94] |
| PIM anticholinergic | 276 (14) | 0.93 [0.55 – 1.55] | 0.9 [0.61 – 1.33] | 0.81 [0.57 – 1.13] |
| PIM cardiovascular | 140 (7) | 0.93 [0.47 – 1.86] | 0.93 [0.55 – 1.57] | 0.82 [0.52 – 1.29] |
| Opioids, not PIM | 645 (32) | 1.36 [0.92 – 2.02] | 0.84 [0.63 – 1.12] | 0.72 [0.56 – 0.92] |
| LOS, ≥ 10 days | 942 (46) | 3.94 [2.67 – 5.81] | 2.34 [1.78 – 3.07] | 2.09 [1.64 – 2.67] |
Note: OR: odds ratio, PIM: potentially inappropriate medication; LOS: length of in-hospital stay. PIM analgesics include dextropropoxyphene and tramadole
Mortality and length of in-hospital stay
The median LOS of the patients was 9 days, with a mean of 9.5 (SD ± 5.7) days, with no significant differences between sexes or age groups, with LOS ≤ 9 days or ≥ 10 days. After adjusting for differences in age, sex and polypharmacy (proxy indicator for comorbidity) patients with a longer LOS (≥ 10 days) had significantly higher six-month mortality (p<0.001), than those with 0-9 days; OR at 30-day 3.94 (95% CI 2.67-5.81), at 90- day 2.34 (95% CI 1.78-3.07) and at 180-day 2.09 (95% CI 1.64- 2.67).
Discussion
In this study, we aimed to improve the knowledge of adverse outcomes in older hip fracture patients exposed to potentially inappropriate medication. Higher mortality rates in hip fracture patients are known to be related to age, male sex, comorbidities, dementia, cancer and cardiovascular diseases [31]. These risk factors are generally unavoidable, hence our focus on the preventable risk factors of potentially inappropriate medication and length of in-hospital stay. In a majority, 81.5%, of the patients who were treated with PIM, we found a correlation between six-month increased mortality and exposure to any PIM, analgesic PIM, and a hospital stay of ten days or longer. National registers with high reliability were used, based on the unique civic number given to all Swedish residents. The statistical examinations were carried out using standard analytical methods, adjusting for identified risk factors that could bias the results.
Lists of PIM for older people have been used ever since the first list was published in 1991 by Mark Beer et al. Since then several up-dated versions have been published, which makes it difficult to compare earlier studies with later ones. Similarly problematic is the use of lists of PIM compiled in other countries, as the drug therapy strategies vary from nation to nation. Consequently we used the Beers’ list from 2015 and added three drugs identified by Swedish health authorities to be inappropriate for older people; propiomazin, tramadole and dextropropoxyphene. Previous studies have shown that treating older people with PIM increase the risk of adverse drug events but other consequences of PIM have not been fully clarified [19,32-35]. As the main results of this study we found frequent prescribing of PIM and analgesic PIM to the patients and associations between higher mortality and old age, male sex, use of any PIM, analgesic PIM and a LOS of ten days or more. The higher mortality associated with old age and male sex shown here, is consistent with results reported in other studies [1,36]. As in previous studies we found that older hip fracture patients are often exposed to PIM, polypharmacy, and cardiovascular drugs [1,12,20,32]. Earlier studies have however not shown any conclusive effect on mortality of PIM and no other study has, to our knowledge, reported such a consistently negative effect on survival. A majority of the study population (60%) was treated with tramadole and dextropropoxyphene, drugs that Swedish health authorities classify as PIM. The lower survival rate shown in this group of patients was mainly associated with exposure to tramadole. These drugs are classified as PIM founded on the frequent incidence of unfavourable adverse reactions in older people such as dizziness, confusion, nausea, fatigue, sweating, muscle cramps and sedation as well as frequent drug interactions. When analysing mortality in patients treated with other types of strong analgesic drugs we found a lower mortality at 180- day, perhaps related to other underlying conditions causing a need for long-term opioid treatment, besides the hip fracture. Dextropropoxyphene was later withdrawn from the Swedish market in 2011 and the prescribing of tramadole has been reduced from representing 27% of all non-opioid prescriptions in the county in 2006, to 11% in 2015.
The results suggest that individually adapted pain management in hip fracture patients is of major importance. Pain is common after osteoporotic fractures and markedly so after a major fracture such as that of the hip [28,29,37]. The consequences of pain are deleterious, especially in older patients, with a high risk of postoperative complications. In a study on hip fracture patients carried out by Morrison et al. in 2002, it was found that higher pain-scores were significantly associated with longer LOS, delayed ambulation and increased functional loss six months after fracture, but with no significant influence on mortality [38]. Previous studies on different methods of pain relief, i.e., epidural analgesia postoperatively, have shown positive effects on ambulation and other functions but not on mortality [7,39,40].
The fact that we could identify higher mortality in relation to a longer LOS, ten days or more, is to some extent in conflict with the results of a longitudinal Swedish study by Nordström et al. [41]. In this study, they found that a break-off point of ten in-hospital days was critical to higher survival after hip fractures, when excluding patients dying during the hospital stay. The result of this study was later discussed upon by Cram and Rush, [42] since a contrasting result was presented by Nikkel et al. on 188, 208 hip fracture patients [43]. In our study, which included deaths occurring in-hospital, there was no significant differences between sexes or age groups associated with the length of hospital stay. It is likely that patients experiencing complications in-hospital require a longer in-hospital stay and that a shorter stay suffices for those without such complications However, both fit patients and patients with multiple comorbidities residing in nursing homes, are discharged after a short period of in-hospital care. It is established from hospital reports that between 35 to 40 per cent of hip fracture patients in the county are residents in nursing homes but it was not within the scope of this study to analyse this further. A longer in-hospital stay can on the other hand present an opportunity for the patients to receive more intense rehabilitation given by professional physiotherapists and thus improve the outcome after a hip fracture.
Included in PIM are many older drugs that were developed before the requirements for premarketing studies were as strict as today. The knowledge of their limitations and risks has mainly been gathered through clinical experience and pharmacological evaluation of the substances. The use of drugs labelled as PIM in treatment of older patients is strongly discouraged, except in the presence of special individual indications. In a 3-year followup study on hip fracture patients, Gosch et al. [18] found that treatment with potentially inappropriate drugs, using the STOPP (Screening Tool of Older Persons’ Prescriptions) criteria, was an independent risk factor for increased long-term mortality but other studies have not shown that effect on mortality from the use of PIM [44].
Limitations and Strengths
There are limitations to this study, both due to missing data on comorbidities as well as residency settings prior to the fracture and after discharge. The absence of data on comorbidities was a disadvantage and for this reason we used polypharmacy as a proxy indicator. Also data on the prescribed and purchased drugs lack the information on whether the drugs were actually consumed or not. We addressed this issue by only using data on drugs that were dispensed more than once during the year. Another limitation was that we did not have access to data on the use of over-the-counter drugs.
The strengths of this study include a clearly defined study population drawn from a large general populace, with no excluded patients, a diagnosis that requires radiographic confirmation and a mandatory database registration for prescribed drugs. The size and range of the population encompass all older hip fracture patients admitted to both university and general hospitals. Residents from rural and urban areas are included and a wide range of prescribing physicians is represented. Therefore the study population encompass a clinically representative group of patients with a hip fracture and the results are likely to be able to generalize upon nationally. The Swedish registers, based on unique civic numbers, provide a reliable source of data with no or very few omitted cases. Swedish pharmacies are by law required to report information on prescription drugs to the national prescription register and are considered to hold robust data. We also tried to adjust for the limitations by using proxy indicators for multiple comorbidity (polypharmacy) and lack of data on drug consumption (at least twice dispensed drugs).
Conclusion
This study proposes to add to the knowledge of potentially avoidable risk factors for mortality in older hip fracture patients. A probable correlation was found between higher six-month mortality and the use of potentially inappropriate medication, especially the analgesic drug tramadol, as well as a hospital stay of ten days or more. The results presented in this study highlight the need for further studies on interventions to reduce adverse outcomes in older hip fracture patients, aiming to optimise their drug treatment, pain management, and in-hospital care.
Further Research
All aspects that can optimise care and treatment of hip fracture patients should be of high priority, as the fractures frequently have an adverse impact on life, function, and cost of care. Consequently, it is crucial that interventions that can potentially lessen these effects are identified. Our results imply that further studies on how to optimise medication, the length of in-hospital stay, and pain management in older patients are required. Increased knowledge of geriatric pharmacology is essential in order to improve positive effects and lessen adverse effects of drug therapy in older people. Interventions to reduce postoperative complications from care-related infections and cardiovascular disease may also have beneficial effects on survival after a hip fracture. Drug reviews by pharmacists specialised in geriatric pharmacology is one way of optimising medication for this group of patients.
Acknowledgements
These works were carried out at the Centre d’Excellence sur le Vieillissement du CHU de Québec (CEVQ). Funds to conduct the studies were granted by Alzheimer Society Canada. A Special Thank to Steve Paquet, research professional at the CEVQ.
Conflict of Interest
The authors have no conflict interest to declare.
Declarations
Ethics approval and consent to participate. Lund University’s Regional Research Board of Ethics approved the study, number 239/2008.
Competing Interests and Funding
The authors declare that they have no competing interests. The project was supported by independent research grants from Skåne County Council’s Research and Development Foundation and Hässleholm Hospital. The grant providers had no role in study design, data collection, statistical analysis, or manuscript preparation and submission. No financial or other support from other sources was received.
Availability of Data and Materials
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Authors’ Contributions
Conception and design of the study: AKE, IA, SE. Data collection: AKE. Statistical analysis and interpretation: AKE, SE, IA. Drafting of the manuscript: AKE, IA, SE. Critical revision of the manuscript and approval of the final version of the manuscript: AKE, IA, SE.
Acknowledgements
The authors would like to thank Mats Pihlsgård, Department of Health, Care and Society, Division of Geriatric Medicine, Lund University, for advice on statistical analyses.
References
- Abrahamsen B, van Staa T, Ariely R, et al. Excess mortality following hip fracture: A systematic epidemiological review. Osteoporos Int. 2009;20(10):1633-50.
- Burge R, Dawson-Hughes B, Solomon DH et al. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res. 2007;22(3):465-75.
- Johnell O, Kanis JA. An estimate of the worldwide prevalence, mortality and disability associated with hip fracture. Osteoporos Int. 2004;15(11):897-902.
- Wiktorowicz ME, Goeree R, Papaioannou A, et al. Economic implications of hip fracture: health service use, institutional care and cost in Canada. Osteoporos Int. 2001;12(4):271-8.
- Kannus P, Parkkari J, Niemi S. Age-adjusted incidence of hip fractures. Lancet. 1995;346(8966):50-1.
- Melton LJ. Hip fractures: a worldwide problem today and tomorrow. Bone. 1993;1:1-8.
- Abou-Setta AM, Beaupre LA, Rashiq S, et al. Comparative effectiveness of pain management interventions for hip fracture: a systematic review. Ann Intern Med, 2011;155(4):234-45.
- Abrahamsen B, Vestergaard P. Declining incidence of hip fractures and the extent of use of anti-osteoporotic therapy in Denmark 1997-2006. Osteoporos Int. 2010;21(3):373-80.
- Bruyere O, Brandi ML, Burlet N, et al. Post-fracture management of patients with hip fracture: a perspective. Curr Med Res Opin. 2008;24(10):2841-51.
- De Rui M, Veronese N, Manzato E, et al. Role of comprehensive geriatric assessment in the management of osteoporotic hip fracture in the elderly: an overview. Disabil Rehabil. 2013;35(9):758-65.
- Zeltzer J, Mitchell RJ, Toson B, et al. Orthogeriatric services associated with lower 30-day mortality for older patients who undergo surgery for hip fracture. Med J Aust. 2014;201(7):409-11.
- Kragh A, Elmståhl S, Atroshi I. Older adults' medication use 6 months before and after hip fracture: a population-based cohort study. J Am Geriatr Soc. 2011;59(5):863-8.
- Kragh Ekstam A, Elmståhl S. Do fall-risk-increasing drugs have an impact on mortality in older hip fracture patients? A population-based cohort study. Clin Interv Aging. 2016;11:489-96.
- Tobias D. Age-related changes in pharmacokinetics and pharmacodynamics: a review. Consult Pharm. 2004;19(8):736-9.
- Trifiro G, Spina E. Age-related changes in pharmacodynamics: focus on drugs acting on central nervous and cardiovascular systems. Curr Drug Metab. 2011;12(7):611-20.
- Walker J, Wynne H. Review: the frequency and severity of adverse drug reactions in elderly people. Age Ageing. 1994;23(3):255-9.
- Veehof LJ, Stewart RE, Meyboom-de Jong B, et al. Adverse drug reactions and polypharmacy in the elderly in general practice. Eur J Clin Pharmacol. 1999;55(7):533-6.
- Gosch M, Wörtz M, Nicholas JA, et al. Inappropriate prescribing as a predictor for long-term mortality after hip fracture. Gerontology. 2014;60(2):114-22.
- Samuelsson KS, Egenvall M, Klarin I, et al. Inappropriate drug use in elderly patients is associated with prolonged hospital stay and increased postoperative mortality after colorectal cancer surgery: a population-based study. Colorectal Dis. 2016;18(2):155-62.
- Thorell K, Ranstad K, Midlöv P, et al. Is use of fall risk-increasing drugs in an elderly population associated with an increased risk of hip fracture, after adjustment for multimorbidity level: a cohort study. BMC Geriatr. 2014;14:131.
- American Geriatrics Society Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63(11):2227-46.
- Griebling TL. Re: American Geriatrics Society 2015 updated beers criteria for potentially inappropriate medication use in older adults. J Urol. 2016;195(3):667-8.
- National Board of Health. Indicators for good drug therapy in the elderly. 2010.
- Berggren M, Stenvall M, Englund U, et al. Co-morbidities, complications and causes of death among people with femoral neck fracture - a three-year follow-up study. BMC Geriatr. 2016;16:120.
- Bliuc D, Nguyen ND, Milch VE, et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301(5):513-21.
- Haentjens P, Autier P, Barette M, et al. Survival and functional outcome according to hip fracture type: a one-year prospective cohort study in elderly women with an intertrochanteric or femoral neck fracture. Bone. 2007;41(6):958-64.
- Dyer SM, Crotty M, Fairhall N, et al. A critical review of the long-term disability outcomes following hip fracture. BMC Geriatr. 2016;16:158.
- Feldt KS, Oh HL. Pain and hip fracture outcomes for older adults. Orthop Nurs. 2000;19(6):35-44.
- Gjertsen JE, Vinje T, Lie SA, et al. Patient satisfaction, pain, and quality of life 4 months after displaced femoral neck fractures: a comparison of 663 fractures treated with internal fixation and 906 with bipolar hemiarthroplasty reported to the Norwegian Hip Fracture Register. Acta Orthop. 2008;79(5):594-601.
- Welfare NBoHa. Indikatorer för god läkemedelsterapi hos äldre. 2010;95.
- Hu F, Jiang C, Shen J, et al. Preoperative predictors for mortality following hip fracture surgery: a systematic review and meta-analysis. Injury. 2012;43(6):676-85.
- Hagstrom K, Nailor M, Lindberg M, et al. Association between potentially inappropriate medication use in elderly adults and hospital-related outcomes. J Am Geriatr Soc. 2015;63(1):185-6.
- Klarin I, Wimo A, Fastbom J. The association of inappropriate drug use with hospitalisation and mortality: a population-based study of the very old. Drugs Aging. 2005;22(1):69-82.
- Juola AL, Pylkkanen S, Kautiainen H, et al. Burden of potentially harmful medications and the association with quality of life and mortality among institutionalized older people. J Am Med Dir Assoc. 2016;17(3):276.
- Hanlon JT, Schmader KE. What types of inappropriate prescribing predict adverse drug reactions in older adults? Ann Pharmacother. 2010;44(6):1110-1.
- Kannegaard PN, van der Mark S, Eiken P, et al. Excess mortality in men compared with women following a hip fracture. National analysis of comedications, comorbidity and survival. Age Ageing. 2010;39(2):203-9.
- Dasch B, Endres HG, Maier C, et al. Fracture-related hip pain in elderly patients with proximal femoral fracture after discharge from stationary treatment. Eur J Pain. 2008;12(2):149-56.
- Morrison RS, Magaziner J, McLaughlin MA, et al. The impact of post-operative pain on outcomes following hip fracture. Pain. 2003;103(3):303-11.
- Foss NB, Kristensen MT, Kristensen BB, et al. Effect of postoperative epidural analgesia on rehabilitation and pain after hip fracture surgery: a randomized, double-blind, placebo-controlled trial. Anesthesiology. 2005; 102(6):1197-204.
- Kehlet H. Effect of postoperative pain treatment on outcome-current status and future strategies. Langenbecks Arch Surg. 2004;389(4):244-9.
- Nordstrom P, Gustafson Y, Michaëlsson K, et al. Length of hospital stay after hip fracture and short term risk of death after discharge: a total cohort study in Sweden. BMJ. 2015;350:696.
- Cram P, Rush RP. Length of hospital stay after hip fracture: how low can we go before patients are at risk? BMJ. 2015;350:823.
- Nikkel LE, Kates SL, Schreck M, et al. Length of hospital stay after hip fracture and risk of early mortality after discharge in New York state: retrospective cohort study. BMJ. 2015;351:6246.
- Gallagher P, O'Mahony D. STOPP (Screening Tool of Older Persons' potentially inappropriate Prescriptions): application to acutely ill elderly patients and comparison with Beers' criteria. Age Ageing. 2008;37(6):673-9.