Research Article - Asian Journal of Biomedical and Pharmaceutical Sciences (2024) Volume 14, Issue 103
Formulation and evaluation of oral tablets using hydroalcholic extracts of Ixora Pavetta
Surya Ravi Kumar1*, Dr. L. Satyanarayana21Department of pharmaceutics, Osmania University, Hyderabad Telangana, India
2Department of Pharmaceutical Biotechnology, Omega college of pharmacy, Ghatkasar, India
- *Corresponding Author:
- Surya Ravi Kumar
Department of pharmaceutics
Osmania University
Hyderabad Telangana, India
E-mail: Suryaravikumar37@gmail.com
Received: 26-Dec-2023, Manuscript No. AABPS-23-123844; Editor assigned: 29-Dec-2023, PreQC No. AABPS-23-123844(PQ); Reviewed: 12-Jan-2024, QC No. AABPS-23-123844; Revised: 17-Jan-2024, Manuscript No. AABPS-23-123844(R); Published: 23-Jan-2024, DOI:10.35841/aabps-14.103.216
Citation: Kumar RS. Formulation and evaluation of oral tablets using hydroalcholic extracts of IXORA PAVETTA. Asian J Biomed Pharm Sci. 2024;14(103):216
Introduction
Herbal medicine has a long and rich history, with diverse traditions spanning across different cultures and civilizations. The use of plants as sources of therapeutic compounds and remedies dates back to ancient times. This article delves into the fascinating world of herbal medicine, highlighting its various traditions and the significance of botanicals in healthcare practices [1].
Traditional Herbal Medicine Traditions
Homemade traditions have played a significant role in shaping the practice of alternative medicine. One notable tradition is the Western homemade convention, which draws from Greek, Roman, and medieval sources. This tradition emphasizes the use of herbs and natural remedies to promote well-being and treat ailments. Additionally, Ayurveda, an ancient healing system from India, emphasizes the holistic approach to health and utilizes herbs in personalized treatments. Another prominent tradition is traditional Chinese medicine, which encompasses various modalities such as acupuncture, herbal formulations, and Qi-based therapies. Chinese herbal medicine, in particular, remains a distinct division of contemporary medical practice [2].
Herbal Medicine in South India
Herbal medicine holds great importance in South India, where it is deeply rooted in the culture and beliefs of the indigenous population. Traditional herbal remedies form an integral part of the healthcare system, providing accessible and affordable options for maintaining well-being. These remedies are often passed down through generations and are used to address a wide range of ailments. They are embraced by the community and offer a holistic approach to health that considers the interconnectedness of the mind, body, and spirit [3].
Botanicals and phytomedicines
Botanicals, commonly referred to as phytomedicines, have always played a vital role in traditional healing practices, especially in developing countries. These natural substances are derived from plants and are known for their diverse chemical compositions with therapeutic properties. In many cultures, botanicals are revered for their ability to support health and wellness. They can be consumed directly as teas or extracts, or they can be incorporated into medicinal preparations such as tinctures, ointments, or capsules [4].
Historical significance and modern applications
The historical significance of herbal medicine cannot be overstated. Many of the drugs used today have their roots in herbal remedies that have been utilized for centuries. Opium, aspirin, digitalis, and quinine are prime examples of substances derived from plants that have been widely used for their medicinal properties. These natural remedies have been refined and standardized over time to meet modern healthcare standards while retaining their effectiveness [5].
The holistic approach
One of the key aspects of herbal medicine is its holistic approach to healing. Traditional systems, such as Ayurveda and Siddha from India, focus on restoring balance and harmony within the body to promote overall well-being. Herbal remedies are often used in conjunction with dietary and lifestyle modifications, as well as mindfulness practices, to support the body's innate healing abilities. This comprehensive approach recognizes that health is a complex interplay of physical, emotional, and spiritual factors [6].
Herbal medicine, with its diverse traditions and emphasis on natural remedies, continues to play a significant role in healthcare practices worldwide. The use of botanicals as sources of therapeutic compounds and remedies has a long history and remains deeply intertwined with cultures and traditions. As we continue to explore the potential of herbal medicine, it is essential to combine traditional knowledge with scientific advancements to ensure safe and effective treatments. By embracing the wisdom of the past and integrating it with modern healthcare approaches, we can harness the power of nature to promote wellness and improve the quality of life. Refining and manipulating plant extracts can enhance their consistency, but the processing methods employed in the pharmaceutical industry may alter their effects in unforeseen ways. In contrast, herbal remedies often contain a complex and diverse blend of chemicals that can provide access to medications or combinations of medicines that have yet to be explored by the pharmaceutical sector. This has led to calls for the regulation of herbal remedies as drugs, ensuring quality standards and demonstrating their efficacy.
The extensive use of herbs in traditional medicine has also raised interest in their potential as preventive and therapeutic agents for various diseases. Natural compounds derived from herbs play a significant role in modern pharmaceuticals. Healthcare professionals and researchers are increasingly turning to medicinal plants for the treatment of conditions such as inflammation, rheumatoid arthritis, cancer, diabetes, and more. These natural remedies are favored due to their minimal side effects, stemming from their organic origins.
To facilitate their administration, herbal extracts are formulated into specific preparations. These novel formulations offer distinct advantages over traditional plant extracts, including improved solubility, enhanced bioavailability, protection against degradation, enhanced pharmacological activity, increased stability, targeted delivery, and resistance to physical and chemical degradation [7].
The development and utilization of these novel herbal formulations hold great potential for advancing medical treatments. By capitalizing on the unique properties and advantages of herbal extracts, researchers aim to optimize their therapeutic potential and provide safer and more effective remedies. However, it is crucial to strike a balance between traditional wisdom and scientific innovation, ensuring that herbal medicines undergo rigorous testing and regulation to guarantee their quality, safety, and efficacy.
In conclusion, herbal remedies offer a rich source of natural compounds with diverse therapeutic properties. While pharmaceutical refinement techniques may alter the effects of plant extracts, herbal remedies provide a complex blend of chemicals that have the potential to unlock new medications and combinations. As the demand for herbal medicines grows, regulations are being implemented to ensure their quality and effectiveness. By exploring the vast potential of herbs, scientists are discovering new ways to address various diseases while minimizing side effects. With careful formulation and research, herbal extracts can be transformed into novel preparations that enhance their bioavailability, stability, and targeted delivery, paving the way for improved healthcare outcomes [8].
Herbal preparations in inflammatory disorders
Inflammatory response might be defense feedback caused by cells harm or damage, identified by redness, heat, swelling, as well as torment. The important objective of inflammation is to center and also kill the aggravation as well as fix the incorporating tissue. Irritability aids the transfer of microorganisms, toxins, or remote textile at the area of damage anticipates their-infect other body organs as well as prepares the area for tissue repair work. This way, it makes a distinction in re-establish tissue homeostasis.1
The restorative plant belongs to the family members Acanthaceae. The significant organically vibrant chemical constituents of the plant are diterpene lactones, either totally free or glycoside. These include andrographolide, deoxyandrographolide, neoandrographolide, andrographolide, andrographolide, etc (Kapil et al., 1993; Trivedi and Rawal, 2000). The corrective effect of the air-borne parts of the plant is spoken to by their in vitro and in vivo anti-hepatotoxic workouts (Gupta et al., 1990; Chander et al., 1995). These thinks about the detailed checked effect of the A. paniculata and its diterpenes andrographolide and also neo andrographolide on the antacid phosphatase (High hill), lotion lipoprotein-X, GPT, GOT, bilirubin but also the influence of A. paniculata on the enterotoxin in animal models. These things furthermore showed up promising defensive effects of the diterpenes andrographolide and neoandrographolide in hepatic injury. In any case, support professional considers with an adequate number of topics is called for to assist show the feasibility of these compounds in individuals2 Various things have actually been executed to explore the corrective impact of this plant. A. paniculata completely expanded the pentoxyresorufin O-dealkylase (Nudge) movement, recommending that A.
Paniculata constituents may affect hepatic cytochrome P-450 chemical.2. In a clinical consideration by Chturvedi et al. (1983) extraordinary comes about by utilizing A. paniculata for the therapy of irresistible hepatitis was completed. Modifications in craving, jaundice, fever, and epigastric distress were seen in all clients within 4 weeks. A significant reduction in lotion bilirubin level (as much as 10-fold) was taped also. Separated from this, High Mountain, SGPT, and SGOT, were as well basically progressed, suggesting that.
Paniculata may well be considered the most inexpensive as well as the primary useful treatment for alluring hepatitis. Although these transpired to show up promising, it should be considered that this ponder carried out more than 3 decades back had mo number of taken into consideration subjects. The requirements for valid control as well as the inadequacy of information from the previous examinations require the transmission of bigger scientific tests to clarify the influences of the dynamic substances. It ought to be considered that in an additional to the major constituents andrographolide as well as Andrographis there might be furthermore various other substances tweaking the bioactivities of the plant. Ultimately, assistance considers the synergistic and opposite influences of the plant substances combined with info about their more extensive atomic instruments are needed to relax the setting of task of A. paniculata. In brief, A. paniculata appears successful in different liver ailments such as viral as well as poisonous liver disease and intra and additional hepatic cholestasis, which would certainly decrease jaundice. All the same, there are too reports nearly its compounds cytotoxicity (Nanduri et al., 2004) and poisonous high quality for the male regenerative structure. Hence, help professional takes into consideration are required to establish the safety as well as professional competence of the long-term use.
Cirrhosis of the liver is the ninth biggest cause of mortality in Western countries, and liver illnesses have a significant worldwide health impact. 1 Although there have been recent advancements in the treatment of chronic viral hepatitis B and C, medications for metabolic, toxic, and genetic liver illnesses are still only moderately effective. The importance of treating advanced cirrhosis, preventing NAFLDs, and other related disorders is frequently overlooked (HCC). With this in mind, it is not unexpected to see the widespread and increasing usage of over-the-counter drugs in patients with liver infections. Certain herbal therapies that slowed the progression of liver fibrosis have been examined in both exploratory and clinical settings, with some now being tested in clinical trials for chronic viral infections. (Figure 1)
UV spectrophotometric method
UV spectrophotometric strategy was created for the in vitro disintegration considers. UV spectrophotometry is an expository procedure utilized routinely for subjective and quantitative measure due the moo taken a toll and unwavering quality amid investigation. (Figure 2)
Plant profile
Ixora pavetta. : It is a plant in the family Rubiaceae that is native to South Asia, especially India, Bangladesh, Sri Lanka and Nicobar Islands²³. It is also known as torch tree, torchwood ixora, torchwood tree, small flowered ixora and other names in different languages¹. It is a shrub or tree that grows up to 10 m tall and has white or pink flowers that are fragrant³⁴. The fruits are globose, black and 2-seeded, and are eaten by sloth bears². The branches of this plant are used for making walls and paste with mud for thatched huts in some villages².
According to some sources, Ixora pavetta has various medicinal uses, such as:
• The fresh leaves juice is used for treating burns, soft tissue wounds, skin infections, malnutrition, pulmonary troubles, liver disorders, hair tonic, sedative, diuretic, diarrhea, dysentery, leucorrhoea and venereal diseases12.
• The roots are bitter, aperient and used for visceral obstructions.
• The decoction of bark is used for anemia, weakness, toothache and to kill worms of mouth and teeth.
• The fomentation made from the leaves is used for hemorrhoid pains3.
Some phytochemical and pharmacological studies have reported that Ixora pavetta has analgesic, anti-inflammatory, anti-ulcer, antioxidant and antimicrobial activities45. However, more research is needed to confirm the safety and efficacy of this plant for human use.
Ixora pavetta, the torch tree, is a plant in the family Rubiaceae. It is a small evergreen tree or shrub, growing to 5-10 m tall. The bark is dark brown and rough. The leaves are opposite, simple, and oblong, measuring 7-15 cm long and 3.5-6.5 cm wide. The flowers are white, fragrant, and borne in corymb-like terminal panicles. The fruits are globose, 2-seeded, and black when ripe [9].
Ixora pavetta is native to South Asia, and is found in India, Sri Lanka, Bangladesh, and Myanmar. It is also cultivated in other parts of the world, including India, Australia, and the Americas.
The plant has a number of medicinal uses. The leaves are used to treat fever, diarrhea, and dysentery. The bark is used to treat ulcers and wounds. The roots are used to treat snakebite and scorpion sting.
Ixora pavetta is also a popular ornamental plant. It is grown for its attractive flowers and foliage. The plant is easy to grow and care for, and can be grown in full sun or partial shade. It is drought tolerant and can tolerate poor soils.
The chemical constituents of Ixora pavetta have been extensively studied. The plant contains a variety of compounds, including
• Flavonoids: These are plant compounds that have antioxidant and anti- inflammatory properties. Some of the flavonoids found in Ixora pavetta include quercetin, kaempferol, and rutin.
• Triterpenoids: These are a type of steroid that have been shown to have anti- inflammatory and anti-cancer properties. Some of the triterpenoids found in Ixora pavetta include ursolic acid, oleanolic acid, and lupeol.
• Tannins: These are plant compounds that have astringent and anti-inflammatory properties. Tannins are found in the bark and leaves of Ixora pavetta.
• Saponins: These are plant compounds that have foaming and detergent properties. Saponins are found in the roots and leaves of Ixora pavetta.
• Alkaloids: These are plant compounds that can have a variety of pharmacological effects, including stimulant, depressant, and hallucinogenic effects. Alkaloids are found in the leaves and flowers of Ixora pavetta.
The chemical constituents of Ixora pavetta have been shown to have a variety of pharmacological activities, including:
• Antioxidant activity: Flavonoids and triterpenoids found in Ixora pavetta have been shown to scavenge free radicals and protect cells from damage.
• Anti-inflammatory activity: Flavonoids, triterpenoids, and tannins found in Ixora pavetta have been shown to reduce inflammation.
• Antibacterial activity: Flavonoids and tannins found in Ixora pavetta have been shown to kill bacteria.
• Antifungal activity: Flavonoids and tannins found in Ixora pavetta have been shown to kill fungi.
• Anticancer activity: Triterpenoids found in Ixora pavetta have been shown to kill cancer cells.
• The chemical constituents of Ixora pavetta have a wide range of potential health benefits. However, more research is needed to confirm these benefits and to determine the safe and effective doses of Ixora pavetta for human consumption.
Here is the taxonomy of Ixora pavetta
• Kingdom: Plantae
• Division: Magnoliophyta
• Class: Magnoliopsida
• Order: Gentianales
• Family: Rubiaceae
• Genus: Ixora
• Species: Ixora pavetta
Ixora pavetta is a member of the family Rubiaceae, which is a large family of flowering plants that includes over 600 genera and 13,000 species. The family is found worldwide, but is most diverse in tropical and subtropical regions. The Rubiaceae family includes a wide variety of plants, including coffee, tea, and gardenias. Ixora pavetta is a small evergreen tree or shrub that can grow to 5-10 meters tall. The bark is dark brown and rough. The leaves are opposite, simple, and oblong, measuring 7-15 centimeters long and 3.5-6.5 centimeters wide. The flowers are white, fragrant, and borne in corymb-like terminal panicles. The fruits are globose, 2-seeded, and black when ripe. Ixora pavetta is native to South Asia, and is found in India, Sri Lanka, Bangladesh, and Myanmar. It is also cultivated in other parts of the world, including India, Australia, and the Americas. The plant has a number of medicinal uses. The leaves are used to treat fever, diarrhea, and dysentery. The bark is used to treat ulcers and wounds. The roots are used to treat snakebite and scorpion sting. Ixora pavetta is also a popular ornamental plant. It is grown for its attractive flowers and foliage. The plant is easy to grow and care for, and can be grown in full sun or partial shade. It is drought tolerant and can tolerate poor soils.There are a number of herbal products that contain Ixora pavetta as an ingredient. Some of these products are available over-the- counter, while others require a prescription from a doctor. Here are some examples of herbal products that contain Ixora pavetta:
• Ayurveda: In Ayurveda, Ixora pavetta is used to treat a variety of conditions, including fever, diarrhea, dysentery, ulcers, wounds, snakebite, and scorpion sting.
• Traditional Chinese Medicine: In Traditional Chinese Medicine, Ixora pavetta is used to treat a variety of conditions, including constipation, diarrhea, dysentery, and jaundice.
• Homeopathy: In homeopathy, Ixora pavetta is used to treat a variety of conditions, including fever, diarrhea, dysentery, and ulcers.
• It is important to note that herbal products are not regulated by the FDA, so it is important to talk to your doctor before taking any herbal product, especially if you have any underlying health conditions.
Aim and Objective
The main aim of the present work is that
This data generation study aims to streamline the process by using natural extracts of IXORA PAVETTA in the production of oral tablets. A 2017 study on rats found that IXORA PAVETTA could help treat atherosclerosis and non- alcoholic fatty liver disease. Affront resistance can arise from either condition or genetics. According to the findings, IXORA PAVETTA reduced liver fatty acid production and affront resistance
Hepatitis B is one of the most common diseases affecting the human population. The initial intergalactic-alpha-alpha treatment is extremely expensive and has a number of negative side effects. Extraction of IXORA PAVETTA has been identified as an alternative homegrown medicine that appears to be beneficial against Hepatitis B and other viral illnesses. As a result, we decided to cultivate IXORA PAVETTA.
Plan of work
The primary objective of the research was to construct a 32-factorial design that might allow the derived plant extract and polymers to achieve a fixed targeted in vitro dissolution profile for controlled delivery for nearly 14 hours.
One of the goals of this project is to create a database for platform selection for an oral controlled release system based on plant extract and polymers for drugs created as herbal remedies, in addition to probe how to simplify it to govern prepared herbal remedies in tablet dosage form.
Literature Review
Ixora pavetta Andrews, (var.: I. Parviflora Vahl.) is a small tree or evergreen shrub belongs to the family Rubiaceae, and is used for many ailments, especially for the treatment of to treat chronic wounds, urinary diseases, skin infection, pulmonary troubles, liver disorder, hair tonic, sedative, diuretic, diarrhoea, dysentery, leucorrhoea and veneral diseases. Preliminary phytochemical screening of ethanol exract of I. pavetta leaf showed the presence of alkaloids, flavonoids, tannins and saponin glycoside. In the analgesic activity, ethanol extract provoked a significant reduction of the number of writhes in acetic acid-induced writhing response, also significantly reduced the licking time in both phases of the formalin test and highest analgesia in hot-plate test (P < 0.01) compared to the control group. The anti- inflammatory effects were investigated employing the both carrageenan and arachidonic acid -induced hind-paw oedema in rats and results of the study revealed the extract to have significant (P < 0.05) anti-inflammatory effects at a dose of 400 and 600 mg/kg p.o., rats in both the models. The antipyretic activity was evaluated using Brewer’s yeast induced pyrexia in rats and the extract showed significant lowering of body temperature in rats at doses of 400 and 600 mg/kg p.o., and no promising results with 200 mg/kg dose level. These findings suggest that ethanol extract of I. pavetta leaves possess potent anti-inflammatory, analgesic and antipyretics [10].
Ixora pavetta Andrews, (var.: I. Parviflora Vahl.) is a small tree or evergreen shrub belongs to the family Rubiaceae. It is commonly grown in India in gardens, Burma and the Andamans. Generally tribes of Paderu division of Visakhapatnam district of Andhra Pradesh, India, used the leaf and roots during chest and muscle pain (1- 4). Similarly, tribes of Nellore district Andhra Pradesh, used root bark infusion as ethnic practice to cures jaundice and burning micturition (5). Flowers is used for Whooping cough, decoction of the stem barks is also used for anaemia and general debility, roots and fruits juice are given to females when urine is highly coloured
Various parts of this plant is also used traditionally in malnutrition, locally to treat-chronic wounds, urinary diseases, skin diseases, pulmonary troubles, Liver disorder, hair tonic, sedative, diuretic, diarrhoea, dysentery, leucorrhoea and veneral diseases (6). Reports on biological activities of the plant are scarce, like ethanolic extract of I. pavetta flower significantly decrease the gastric secretion in the aspirin induced and pylorus ligated rats (5). However, only a few phytochemical have been reported on this plant in the literature like ixoral, beta-sitosterol, rutin and kaempferol-3-rutinoside was isolated from leaves and flower; Ethanolic extract of I. pavetta leaves also contain 3-Butyn-2-ol, 3-Butyn-1-ol, amyl nitrite, 2-Octyn- 1-ol, 1, 9-Decadiyne and Butyl glyoxylate. Stems gave a flavoneglycoside, chrysin 5-O-beta-D-xylopyranoside (6). The arial parts contain 6, 7-dimethoxycoumarin and the seed oil gave capric, lauric, myristic, palmitic, stearic, arachidic, behenic, oleic and linoleic acids.
Kiritkar KR et al1 and Thabrew MI2 et al reported that the plant leaves were used in the treatment of liver diseases, pain of piles, urinary diseases and fever. Ethanol extract of P.Indica leaves was obtained by the extraction procedure as described by Chattopadhyay3 with slight modification.Powder leaves was defatted with petroleum ether(60-80 c) and Soxhlet extraction with 99.9% ethanol.Further the ethanol extract was distilled and solvent ethanol removed. The residue extract was dried and measured. The yield was 28 gm. The extract thus obtained was used as the study material in the entire study for its hepatoprotective activity in albino rats.Various phytochemical constituents like flavonoids and their glycosides, alkaloids, sterols, phenolics, lignins, terpenoids, coumarins, fatty acids, saponins have been isolated from this plant.
Methodology
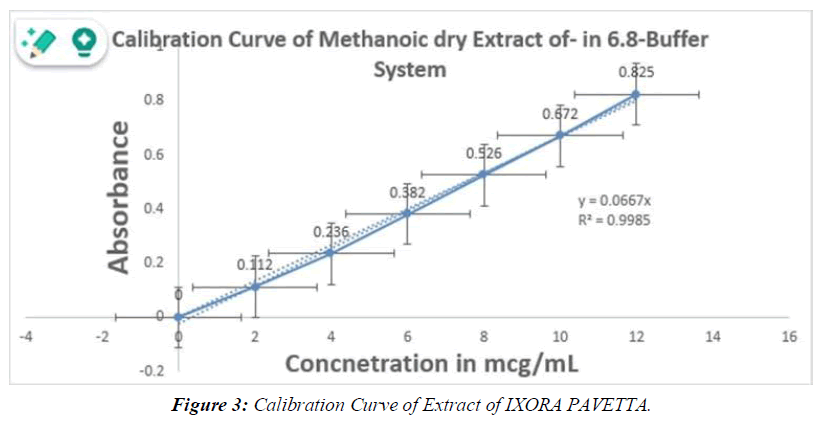
Method for Making preparations Stock Solutions and Serial Dilutions: To render this same stock solution, dissolve 100 mg of IXORA PAVETTAworking standard extract in 6.8 buffer and liquefy to 100 ml with water. For subsequent serial dilutions, the 6.8 buffer was used. (Table 1, Figure 3)
| Concentration (mcg/ml) | Absorbance |
|---|---|
| 0 | 0 |
| 2 | 0.112 |
| 4 | 0.236 |
| 6 | 0.382 |
| 8 | 0.526 |
| 10 | 0.672 |
| 12 | 0.825 |
Table 1: Calibration Curve of Extract of IXORA PAVETTA
Preformulation
Choice of Polymers
Initially three polymers were chosen Sodium CMC (Carboxy Methyl Cellulose)- medium consistency, Ethyl-Cellulose 7cps, IXORA PAVETTARL-100. Each polymer was made use of separately for the meaning. The resolution of the polymers was based upon the appropriate detailing of topical polymeric. For the primary screening of the polymer the work was based upon Schroeder et al. At first the assessment framework was based upon 3 requirements: self- glue, cosmetically appeal as well as drying time. The self-adhesive building of the shaping polymer is called for to be high adequate so as to empower an application to be linked straightforwardly, which would certainly ensure most practical for the understanding. The shaped is needed to be non-sticky from the back so regarding dodge grip to the outfit of the understanding or various other physical get in touch with. Thus a backing layer was produced of PVA. Taking into consideration the reality that various clients whine approximately the high perceivability of transdermal patches which is thought about cosmetically unsightly the polymeric is thought to be straightforward. Drying time was taken into thought. The interpretation should certainly take the very least time to make. In development to this, the transportation structure is called for to appear specific continuous high quality on the skin and also nail in order to have the ability to provide a continuously sedate supply over an extracted amount of time. Dissolvable casting approach was used for the arrangement of the topical. Each polymer was used separately [11].
FTIR studies for extracts
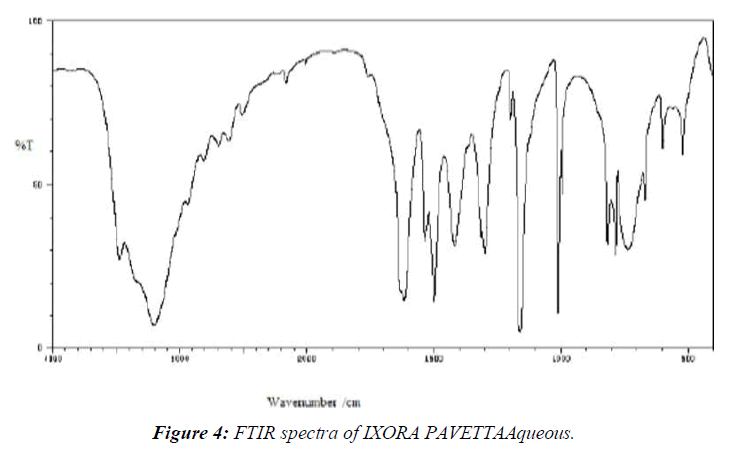
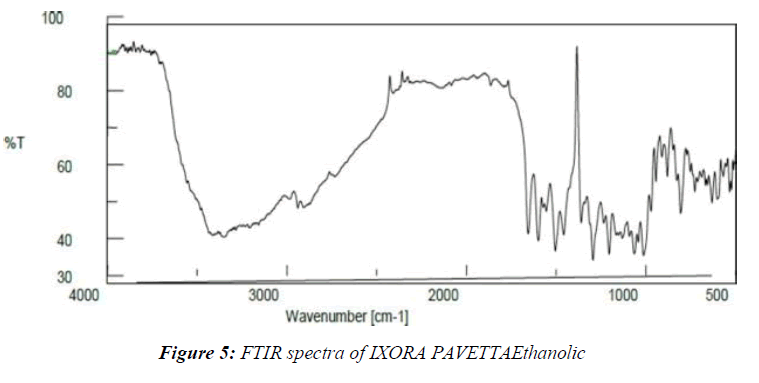
The IR spectra were captured using an FTIR spectrophotometer (Jasco-450, also from Japan) and just a diffuse reflectance guideline. Blending this same test with KBr, triturating in a glass mortar, and finally establishing within the test holder all were part of the test planning. The blends were created by triturating the sedate with the excipient and storing them for 24 hours at room temperature in a tightly closed vial. The spectra were explored over a frequency range of 400 - 400 cm-1. (Table 2-4, Figure 4,5)
| Frequency (cm-1) | Assignment |
|---|---|
| 3332 | OH stretch |
| 2921 | CH stretch |
| 1662 | C=O |
| 1621 | C=C |
| 1601 | Aromectic Structure |
| 1512 | C=C |
| 1361 | C-O-C |
| 1296 | C-O-C |
| 1201 | C-O-C |
| 1261 | C-O-C |
| 813 | Substituted Aromectics |
Table 2: Interpretation of IR Spectra for IXORA PAVETTAAqueous
| Frequency (cm-1) | Assignment |
|---|---|
| 3332 | OH stretch |
| 2924 | CH stretch |
| 1661 | C=O |
| 1621 | C=C |
| 1601 | Aromectic Structure |
| 1512 | C=C |
| 1361 | C-O-C |
| 1296 | C-O-C |
| 1201 | C-O-C |
| 1061 | C-O-C |
| 813 | Substituted Aromectics |
Table 3: Interpretation of IR Spectra of IXORA PAVETTAEthanolic extract
| Factors | Levels | |||
|---|---|---|---|---|
| -1 | 0 | +1 | ||
| X 1 | Concentration of the Extract of IXORA PAVETTA | 450mg | 500mg | 550mg |
| X 2 | Concentration of PEG | 0.1 ml | 0.15ml | 0.2ml |
Table 4: Coded levels translated in actual units.
Formulation of IXORA PAVETTA, Ethyl cellulose
HHH 15ml ethanol was placed in a 50ml measuring utensil, followed by propylene glycol, glycerol, and PEG 400 after proper blending. As a result, a distinct arrangement emerged. The extricates were then incorporated into this clear arrangement, and the sedate was broken down again, yielding a clear arrangement. The polymer was then progressed in this configuration and became solubilized. The obtained arrangement was then placed in the petri- dish that already contained the backing membrane and dried in the open discussion.
3² Full factorial Experimental Design Layout
Independent variable:
X1= Concentration of IXORA PAVETTARL 100 X2= Concentration of PEG 400 (ml)
Dependent variable:
Y1= In-vitro Drug Release (%)
Y2= Adhesive Strength (g/20mm)
Factorial batches of formulations (Table 5)
| Sl. No | Batch No | X1 | X2 | Conc of Formingagent (inmg) | Cone of PEG (inml) |
|---|---|---|---|---|---|
| 1 | F1 | 0 | 0 | 500 | 0.15 |
| 2 | F2 | 0 | -1 | 500 | 0.1 |
| 3 | F3 | 0 | 1 | 500 | 0.2 |
| 4 | F4 | -1 | 0 | 450 | 0.15 |
| 5 | F5 | -1 | -1 | 450 | 0.1 |
| 6 | F6 | -1 | 1 | 450 | 0.2 |
| 7 | F7 | 1 | 0 | 550 | 0.15 |
| 8 | F8 | 1 | -1 | 550 | 0.1 |
| 9 | F9 | 1 | 1 | 550 | 0.2 |
Table. 5: Factorial design layout.
Independent variable
X1= Concentration of the Extract of IXORA PAVETTA
X2= Concentration of PEG 400 (ml) Dependent variable:
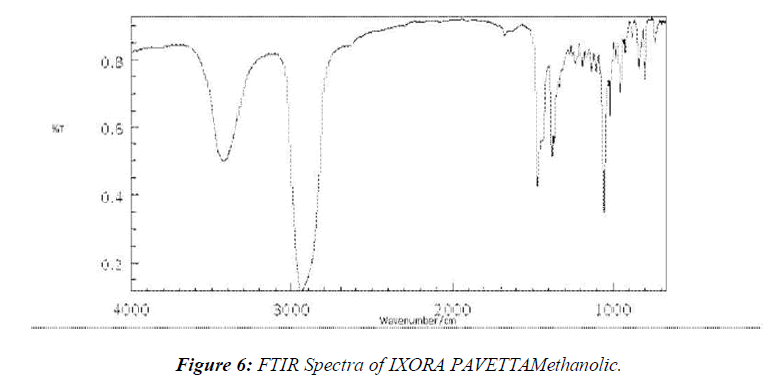
Y1= In-vitro Drug Release (%) Y2= Adhesive Strength (g/20mm) (Table 6, 7, Figure 6)
| Sr. No | Name of ingredient | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | IXORA PAVETT A(mg | 500 | 500 | 500 | 450 | 450 | 450 | 550 | 550 | 550 |
| 2. | HPMC K 4M(mg) | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 |
| 3 | Aerosil | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| 4 | Mg.Stearate | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| 5 | PEG 400 (ml) | 0.15 | 0.1 | 0.2 | 0.15 | 0.1 | 0.2 | 0.15 | 0.1 | 0.2 |
| 6. | Oleic acid(ml) | 0.075 | 0.075 | 0.075 | 0.075 | 0.075 | 0.075 | 0.075 | 0.075 | 0.075 |
| 7. | Talc | 15 | 15 | 15 | 15 | 15 | 15 | 15 | 15 | 15 |
| 8. | Ethanol | q.s | q.s | q.s | q.s | q.s | q.s | q.s | q.s | q.s |
Table. 6: Actual factorial design batches of IXORA PAVETTAextracts
| USP | Max % difference allowed | IP/BP |
|---|---|---|
| 130mg> or less | 10% | 80mg>or less |
| 120mg-324mg | 7.50% | 80mg-250mg |
| 324mg< or more | 5% | 250mg<or more |
Table 7: IP, USP and BP standards of percentage of weight variation
Evaluation Parameters
Physical Appearance
The sample's physical appearance was witnessed. The sample's colour, appearance, proximity to any clogging/precipitation, and surface were all evaluated.
Thickness
The thickness of the sample was measured with a micrometer screw gauge at three distinct spots, as well as the cruel esteem was calculated.
Weight Change
Weight variation was considered by weighing only three chosen circular strands of range from each arrangement. This was accomplished for each definition, and cruel esteem was calculated. (Table 8)
| Sl. No | Evaluation Parameter | Mean + Std Dev (n=3) for of Phyllanthus Niruri |
|---|---|---|
| 1 | Foreign Matter | 1.38 |
| 2 | Moisture Content | 10.42 |
| 3 | Total Ash Value | 13.72 |
| 4 | Water Soluble Ash Value | 3.12 |
| 5 | Acid Insoluble Ash Value | 12.3 |
| 6 | Water Soluble Extractive Value | 5.64 |
| 7 | Chlorofrom Soluble Extractive Value | 0.16 |
| 8 | Methanol Soluble Extractive Value | 6.47 |
| 9 | Ethanol Soluble Extractive Value | 5.23 |
Table 8: Determination of physical constants.
Moisture uptake study
At 25 degrees Celsius, a sample was left in a desiccator with silica gel for 24 hours, weighed (Ws), and then relocated to another desiccator with soaking NaCl solution (relative mugginess: 75%). Once harmony was attained, the layer was removed and weighed (Wm). The capacity to absorb moisture was calculated using the following instances:
Drug content
A small amount of sample was added to a container containing 100 mL of methanol for sedate substance assurance. It then stirred for 4-5 hours with the help of an attractive stirrer. The arrangement was then tested for sedate substance at every discovery wavelength.
Adhesive Test
The patches were placed in strips 20 mm wide and 40 mm long, and they were condition for 24 hours at 23 °C and 50% RH. The tests were mounted on a stainless steel discipline plate and smoothed five times with a 4.5-pound roller. Each attached test's lower conclusion was tied with string. The Ubique Model- UTT-P computerized malleable quality analyzer was used to perform the test on a stainless steel plate. The plate was placed vertically on the lower clamp, and the string was fastened to the upper clamp and pulled away from the substrate at a speed of 300 mm per minute at a temperature of 180°C. The stainless steel plate employed for the test was installed on the lower clamp of the Ubique Computerized Ductile Quality Analyzer, Model-UTT-P, and the cement was run. The peel adhesive test was calculated in g/20mm.
In vitro drug release study
In vitro medicate discharge Pondering was done in a Franz dissemination cell with a receptor segment capacity of 25 ml of phosphate buffer at pH 7.2 for IXORA PAVETTAlayer and pH 6.8 for Spathodea campanulata layer. Cellophane dialysis that had been soaked in separate buffer solutions for 24 hours. To ensure that the topical layer released a sedate, the sample's estimated pore size of 2.4 nm was used. The substance in the receptor compartment was blended at 50 rpm while thermostatically balanced to 370.5 °C using an appealing stirrer. A compartment and a topical layer were joined. After 1, 2, 3, 4, and 8 hours, the testing (2 ml) was stopped. Having followed each examination, a split even with volume of new Phosphate buffer was promptly added to the receptor cell. For IXORA PAVETTA, the drug concentration was determined spectrophotometrically at 281 nm.
Stability studies
Stability tests on the optimized layers of Forum and SCME were performed in accordance with the Criteria. The layer was stored in an aluminum thwart and subjected to elevated temperature and stickiness conditions of 40 20C and 75 5% RH, respectively. At the end of 0, 30, 60, and 90 days, 120, 150, and 180 days, tests were pulled back and judged for adhesive and pliable quality.
Stability studies were also performed on product 37°C ± 2°C at 75 ± 5% RH IXORA PAVETTA optimized were exposed to various temperature conditions in a cyclic design that retells the changes likely to be experienced during its use or in the dispersion handle. During the course of the investigation, the picked details were confined to refrigerated temperature (2 - 8° C) for two days, pursued by 40° C for two days, one cycle. Three cycles were completed in twelve days, and significant changes were seen. All through stability studies, the chosen gels also were placed under the following temperature and relative mugginess situations. For six months, formulations were evaluated for various parameters at the end of each month. The pH, appearance, and spreading coefficient of the — widely were all assessed.
Results and Discussion
Evaluation of Physical constants
Physical constants were decided for the powdered rhizomes. The experiments were done in triplicate, and the results are listed below. (Table 9)
| Sl. No | Name of Plant | Type | Colour of ectract | Appearnce | % Yield | ph |
|---|---|---|---|---|---|---|
| 1 | Hydrophilia auriculate | Aqueous | Reddidh brown | Extricates | 4 | 5 |
| 2 | Ethanolic | Reddidh brown | Extricates | 8.3 | 6-May | |
| 3 | Mithanolic | Reddidh brown | Extricates | 14.4 | 5 |
Table. 9: Properties of extracts
The initial examination produced favorable results for exterior matter, moisture substance, cinder value, and extractive values, as shown in the table above. Aspidium cicutarium had a water solvent extractive value of 5.69% and a methanolic extractive worth of 6.47%. The increased methanolic extractive suggests that methanolic extract could be used to help with phytochemical analysis. Spathodea campanulata garnered preferable results in terms of distant matter, moisture substance, fiery debris value, and extractive properties. The methanolic extractive esteem was 2.09 percent, while the aqueous solvent extractive esteem was 3.73 percent. This leads us to believe that extraction can be fulfilled using methanol and water.
Extraction
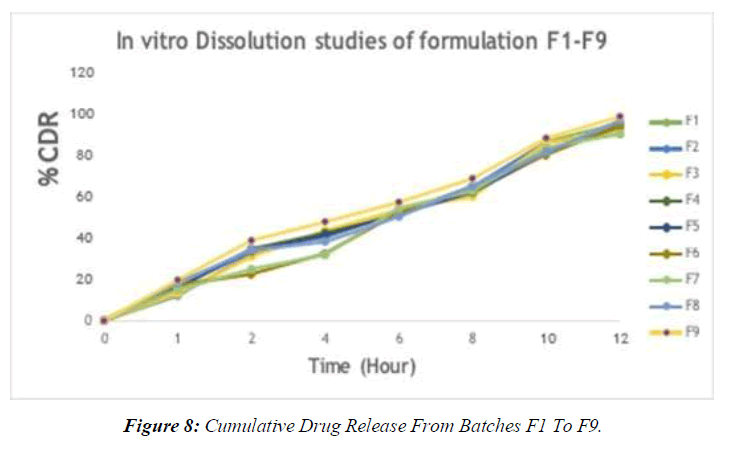
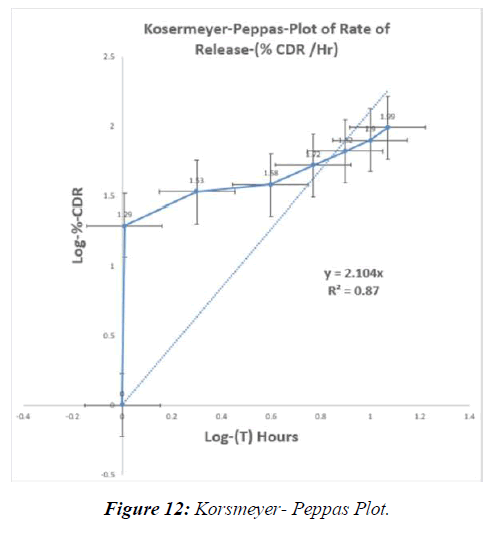
The powdered rhizomes of IXORA PAVETTA were extracted using three different solvents. The following are the properties of plant solvent extracts. IXORA PAVETTAextracts produced marginally sticky green extricates in all instances. The table below shows the properties of extracts and phytochemical enquiry. (Table 10-14, Figure 7-12)
| F. No | Bulk density (gm/cc) | Tapped density (gm/cc) | Angle of repose | Carr’s index | Hausner’s ratio |
|---|---|---|---|---|---|
| F1 | 0.272 | 0.335 | 18.43 | 11.62 | 1.11 |
| F2 | 0.254 | 0.376 | 25.46 | 23.74 | 1.32 |
| F3 | 0.247 | 0.345 | 21.32 | 20.35 | 1.24 |
| F4 | 0.288 | 0.339 | 21.43 | 18.46 | 1.28 |
| F5 | 0.243 | 0.343 | 22.17 | 19.34 | 1.24 |
| F6 | 0.237 | 0.337 | 20.13 | 16.12 | 1.16 |
| F7 | 0.239 | 0.384 | 21.43 | 18.64 | 1.22 |
| F8 | 0.235 | 0.383 | 22.28 | 19.16 | 1.23 |
| F9 | 0.283 | 0.398 | 20.35 | 19.41 | 1.36 |
Table. 10: Precompression parameters F1- F9
| F. No. | Angle of repose | Carr’s index | Hausner’s ratio |
|---|---|---|---|
| F1 | 18.43 | 11.62 | 1.11 |
| F2 | 25.46 | 23.74 | 1.32 |
| F3 | 21.32 | 20.35 | 1.24 |
| F4 | 21.43 | 18.46 | 1.28 |
| F5 | 22.17 | 19.34 | 1.24 |
| F6 | 20.13 | 16.12 | 1.16 |
| F7 | 21.43 | 18.64 | 1.22 |
| F8 | 22.28 | 19.16 | 1.23 |
| F9 | 20.35 | 19.41 | 1.36 |
Table 11: Evaluation of adhesive strength, moisture uptake and drug content from F1 to F9
| Time in hours | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 15.9 | 12.3 | 13.2 | 15.8 | 16.6 | 17.3 | 15 | 18.6 | 19.9 |
| 2 | 35.2 | 32.8 | 31.3 | 34.9 | 33.8 | 22.6 | 25 | 34.5 | 39.2 |
| 4 | 40.2 | 41.2 | 43.8 | 42.7 | 41.5 | 32.6 | 32 | 38.5 | 48.3 |
| 6 | 53.3 | 52.5 | 53.9 | 51.4 | 51.9 | 52.4 | 55 | 50.8 | 57.9 |
| 8 | 62.5 | 65.5 | 60.4 | 64.4 | 62.5 | 65.1 | 63 | 65.9 | 69.5 |
| 10 | 87.6 | 87.4 | 86.6 | 82.5 | 81.3 | 80.8 | 85 | 82.5 | 89.2 |
| 12 | 96.6 | 91.3 | 92.2 | 94.4 | 95.3 | 94.9 | 91 | 97.9 | 99.8 |
Table 12: % Cumulative Drug Release from batches F1 to F9
| Time (Hour) | % CDR | % ARR | Log % ARR | Log % CDR | √t | Log t |
|---|---|---|---|---|---|---|
| 1 | 19.6 | 80.4 | 1.90 | 1.29 | 1 | 0 |
| 2 | 34.2 | 65.8 | 1.81 | 1.53 | 1.414 | 0.30 |
| 4 | 38.3 | 61.7 | 1.91 | 1.58 | 2 | 0.60 |
| 6 | 52.9 | 47.1 | 1.79 | 1.72 | 2.44 | 0.77 |
| 8 | 66.5 | 33.5 | 1.52 | 1.82 | 2.82 | 0.90 |
| 10 | 80.2 | 19.8 | 1.29 | 1.90 | 3.16 | 1 |
| 12 | 99.8 | 0.2 | 0.06 | 1.99 | 3.46 | 1.07 |
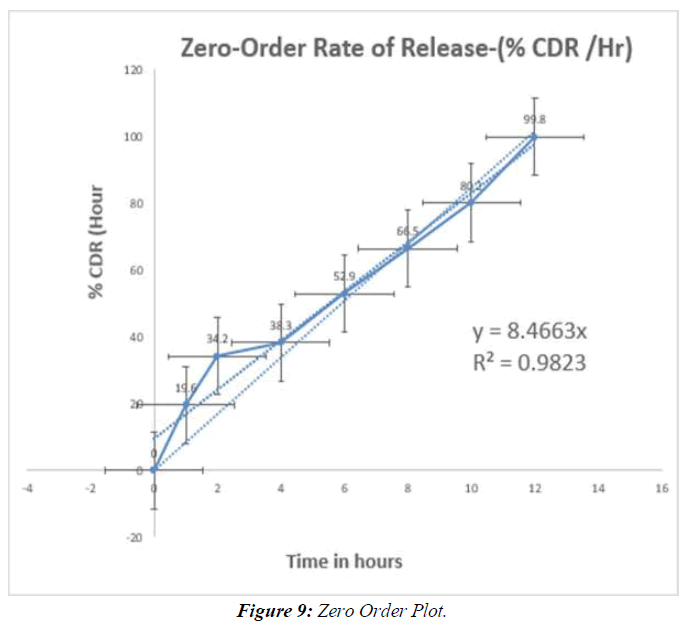
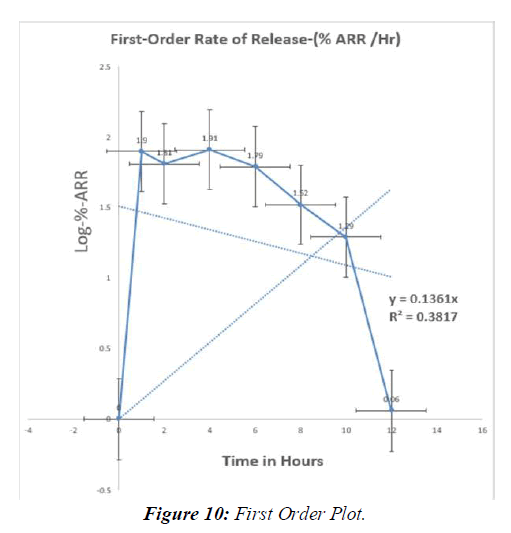
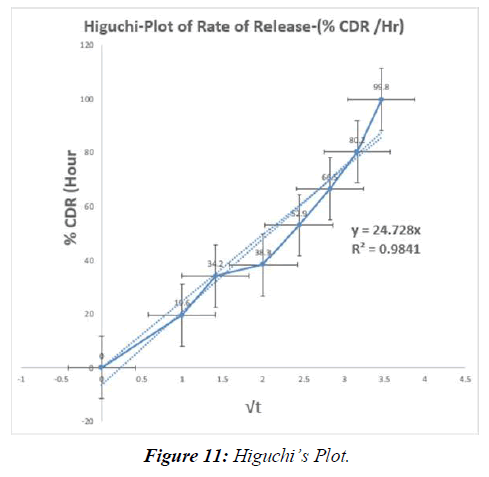
Tabl. 13: In-vitro drug release kinetic studies of optimised formulation (F9)
| Formulation | R2value of Zeroorder | R2value of 1st order | R2value of Higuchi model | R2value of Peppa’s model | ‘n’ value of Peppa’s model |
|---|---|---|---|---|---|
| F9 | 0.9852 | 0.1347 | 0.9852 | 0.6621 | 0.1847 |
Table 14: Regression values of in-vitro release kinetic study optimized (F9)
Summary and Conclusion
To summarize, the contents of the IXORA PAVETTAherbal extract, also known as Bhumi Amla, were extracted using chemical liquids including aqueous, ethanolic, and methanolic solutions. The FTIR extracts obtained all had distinct peaks. A 32 factorial design was used to create the tablets. pre- compression. The F9 formulation outscored all others in terms of medication release percentage and pre-compression outcomes. The invitro release kinetic analysis of the F9 formulation revealed that the formulation's "n" value was 0.0992 when we executed an adhesive strength test for post formulation parameters. This demonstrates that the medicine is dissolving as the formulation design intended.
References
- Latha LY, Darah I, Jain K, Sasidharan S (2012) Pharmacological screening of methanolic extract of Ixora species. Asian Pacific J Tropic Bio Medici. 2: 149-151.
- Srinivas K, Celestinbaboor V (2011) Antiulcer activityof Ixora pavetta, Internati J Current Pharmace Res. 3: 1-2.
- Mahato RI, Narang AS. (2011) Pharmaceutical Dosage Forms and Drug Delivery. Drug Deliver Systems, 217-34.
- Parul S, Anoop K, Pankaj S, et al (2012) Fast disintegrating oral films: A recent trend of drug delivery. Int J Drug Dev Res. 4:80-94.
- Reddy PD, Swarnalatha D. (2010) Recent advances in novel drug delivery systems. Int J Pharm Technol Res. 3:2025-7.
- Rastogi S, Vaya N, Mishra B (1995) Osmotic pump: A novel concept in rate controlled oral drug delivery. East Pharm, 38:79-89.
- Pahwa R, Piplani M, Sharma PC, et al (2010) Orally disintegrating tablets-Friendly to pediatrics and geriatrics. Arch Appl Sci Res.
- Hirani JJ, Rathod DA, Vadalia KR (2009) Orally disintegrating tablets: A review. Trop J Pharm Res. 8:161-72.
- Ishikawa T, Mukai B, Shiraishi S, et al (2001) Preparation of rapidly disintegrating tablet using new types of microcrystalline cellulose (PH-M series) and low substituted-hydroxypropylcellulose or spherical sugar granules by direct compression method. Chem Pharm Bull (Tokyo) 49:134-9.
- Ghosh T, Ghosh A, Prasad D (2011) A review on new generation orodispersible tablets and its future prospective. Int J Pharm Pharm Sci. 3:1-7.
- Preethi S, Padmapriya S, Rajalakshmi AN (2019) Review on Dispersible Tablets: A New Endeavor in Drug Delivery Syste. nl J Pharm Biol Sci 9:1209-1222
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref