Short Communication - International Journal of Pure and Applied Zoology (2022) Volume 10, Issue 11
ECOLOGICAL STATUS OF WATER LAKE (KANMAI) IN AND AROUND KADAICHANENTHAL AT MADURAI DISTRICT, TAMIL NADU, INDIA: A PRELIMINARY STUDY
Selvaraj selvamurugan*
Department of Clinical Development Service Agency, Translational Health Science and Technology Institute Building (THSTI), Faridabad-121001, India
- Corresponding Author:
- Selvaraj Selvamurugan
Department of Clinical Development Service Agency
Translational Health Science and Technology Institute Building (THSTI)
Faridabad-121001, India
E-mail: selva199420@yahoo.in
Received: 13-Oct-2022, Manuscript No. IJPAZ-22-76928; Editor assigned: 14-Oct-2022, PreQC No. IJPAZ-22-76928(PQ); Reviewed: 28-Oct-2022, QC No. IJPAZ-22-76928; Revised: 03-Nov-2022, Manuscript No. IJPAZ-22-76928(R); Published: 10-Nov-2022, DOI: 10.35841/2320-9585-10.11.154
Abstract
In present study carried out the butterflies’ 34 species and 5 families, Birds 24 species, 5 order and 13 families and Dragonfly 13 species and 3 families is recorded. The present study location is also facing tremendous conservation challenges by the impact of anthropogenic alteration of the habitats in and around the lake. In this paper showing a baseline data of butterfly, birds and Odonatas.
Keywords
Lepidoptera, Odonata, Avifauna, Diversity, Lake Ecology, Wetlands.
Introduction
Aquatic ecosystems are important one which provide livelihoods for the millions of people who live around them. Man depends ponds for most of his needs like fishing, agriculture, irrigation, and other domestic purposes. Ponds are playing a very good role in rain harvesting, storage of water and regulation of ground water level. So in order to maintain the ground water level we must conserve ponds and pond habitat [1]. The order Lepidoptera is divided into two suborders viz., Heterocera (Moths) and Rhopalocera (Butterflies). So far, about 1,57,424 species of Lepidoptera have been described globally [2]. There are about 18,000 species of butterflies in the world and India has 1,501 species of butterflies [3]. The Western Ghats harbours around 330 species of butterflies. The main causes for the decline of butterfly populations are deforestation, habitat destruction for urbanization, industrialization and agriculture causes changes in temperature, humidity and rainfall. Prevalence of unfavorable weather conditions often affect habitat suitability leading to local extinction of butterflies. Unfortunately developmental activities and resulting habitat fragmentation create threats to the survival of butterflies worldwide.
India is a home of many species of birds including local as well as migrant birds. Water birds are ecologically dependent on wetlands. They play an important role in human life on culturally, socially, scientifically and as a food resource. The Indian subcontinent supports diverse avifauna (1370 species i.e. 13% of the world’s birds) and which includes 141 endemic species [4]. Tamil Nadu is known for its rich diversity of avifauna with more than 450 species including several endemic and conservation prioritized species [5]. Wetlands and water birds are inseparable elements; wetlands are serve as a reservoir for sustaining native flora and fauna [6]. The aquatic birds are important bio-indicators of lake ecosystems which should be protected to conserve the biodiversity and environment [7].
Wetlands in India face tremendous anthropogenic pressure mainly due to the release of domestic sewage, industrial effluents, dumping of solid waste, over-exploitation of the natural resources and conversion of wetlands into barren lands. This resulted in biodiversity loss and disturbance of the wetland services [8]. This loss of wetlands has dangerously reduced the availability stopover sites for migrating birds and has increased the importance of remaining wetlands to migrants as well as nesting species.
Odonates are playing an important role in climate change mainly due to evolutionary history and its adaptations. Odonata (dragonflies and damselflies) are gorgeous aquatic insects distributed throughout the world. Globally, 5952 species of Odonates under 652 genera have been reported. In India, 474 species and 50 subspecies which belongs to 142 genera and 18 families were recorded [9].
The aim of current study is to find out the current status of butterflies, birds and Odonatas in kadaichanenthal lake and The study was also intended to generate baseline reference data to evolve a suitable for Healthy ecosystem.
Study area
Kadachanendhal (9.9248537, 78.1450406) is a Locality in Madurai West City in Tamil Nadu State, India (Figure 1). Showing the lake of kadaichanenthal (Figure 2). Showing the growing of weeds and lotus. Butterflies and Birds were primarily identified directly in the field by observation and use of the field guide. The difficult cases followed capture or photography of the species. This wetland provides water for fishing activities or aquacultural practice as well as irrigation to surrounding agricultural lands.
Results and discussion
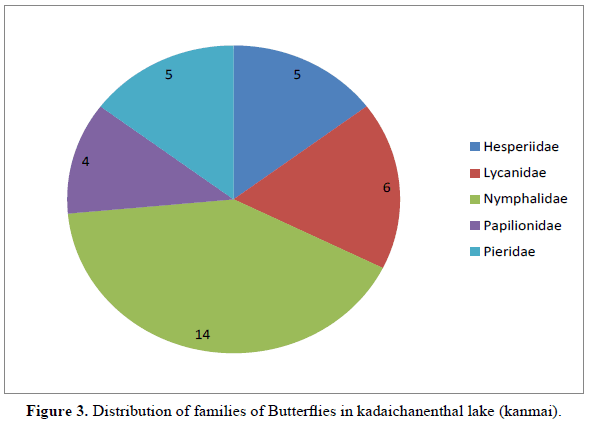
A total of butterflies 34 species and 5 families were recorded from the study area (Table 1). The family is are Hesperiidae (5 species), Lycanidae (6 species), Nymphalidae (14 species ), Papilionidae(4 species), Pieridae (5 species) (Figure 3). In many ecologically stable communities, plants have a strong role in the determination of the structure of the community and the eventual faunal survival in it [10]. The growth rate of Lepidopteran individuals depend on the host plants with constitute the nutritional composition of the insects. For butterflies, it is important to have sufficient food sources for caterpillars and adults. We need to focus on species diversity and representation of flowering plants in the habitat. Localities should have a minimum size (which is different for each species) and should not be too far apart from each other to prevent populations from isolation. Only like that they will be able to function properly and maintain the necessary number of individuals and genetic diversity, which is necessary for theirs sustainable development [11].
| S.No | Scientific name | Common name | Family |
|---|---|---|---|
| 1 | Aeromachus pygmaeus Fabricius | Pygmy Scrub Hopper | Hesperiidae |
| 2 | Amittia dioscorides Fabricius | Bush Hopper | Hesperiidae |
| 3 | Badamia exclamationis Fabricius | Brown Awl | Hesperiidae |
| 4 | Baoris farri Moore | Paintbrush swift | Hesperiidae |
| 5 | Borbo cinnara Wallace | Rice Swift | Hesperiidae |
| 6 | Lampides boeticus Linnaeus | Pea Blue | Lycanidae |
| 7 | Talicada nyseus Guerin-Meneville | Red Pierrot | Lycanidae |
| 8 | Zizina otis Fabricius | Lesser Grass Blue | Lycanidae |
| 9 | Castalius rosimon Fabricius | Common Pierrot | Lycanidae |
| 10 | Chilades lajus Stoll | Lime Blue | Lycanidae |
| 11 | Discolampa ethion Westwood Banded | Blue Pierrot | Lycanidae |
| 12 | Euploea core Cramer | Common Crow | Nymphalidae |
| 13 | J. atlites Linnaeus | Grey Pansy | Nymphalidae |
| 14 | Danaus chrysipppus Linnaeus | Plain Tiger | Nymphalidae |
| 15 | J. iphita Cramer | Chocolate Pansy | Nymphalidae |
| 16 | J. hierta Fabricius | Yellow Pansy | Nymphalidae |
| 17 | J. orithiya Linnaeus | Blue Pansy | Nymphalidae |
| 18 | Lethe drypetis Hewitson | Tamil Tree Brown | Nymphalidae |
| 19 | L. rohria Fabricius | Common Tree Brown | Nymphalidae |
| 20 | Melanitis leda Linnaeus | Common Evening Brown | Nymphalidae |
| 21 | M. zitenius Herbst | Great Evening Brown | Nymphalidae |
| 22 | Moduza procris Cramer | Commander | Nymphalidae |
| 23 | Mycalesis patina Moore Gladeye | Bush Brown | Nymphalidae |
| 24 | Phalanta phalantha Drury | Common Leopard | Nymphalidae |
| 25 | Neptis hylas Linnaeus | Common Sailer | Nymphalidae |
| 26 | Belenois autrota Fabricius | Pioneer | Pieridae |
| 27 | Catopsilia pyranthe Linnaeus | Mottled Emigrant | Pieridae |
| 28 | Delias eucharis Drury | Common jezebel | Pieridae |
| 29 | Hebomoia glaucippe Linnaeus | Great Orange Tip | Pieridae |
| 30 | Leptosia nina Fabricius | Psych | Pieridae |
| 31 | G. doson C.&R. Felder | Common Jay | Papilionidae |
| 32 | P. memnon Linnaeus | Blue Mormon | Papilionidae |
| 33 | Papilio demoleus Linnaeus | Lime Butterfly | Papilionidae |
| 34 | Atrophaneura aristolochiae Fabricius | Common Rose | Papilionidae |
Table 1. Butterfly diversity of water lake (kanmai) kadaichanenthal at Madurai district, Tamil Nadu.
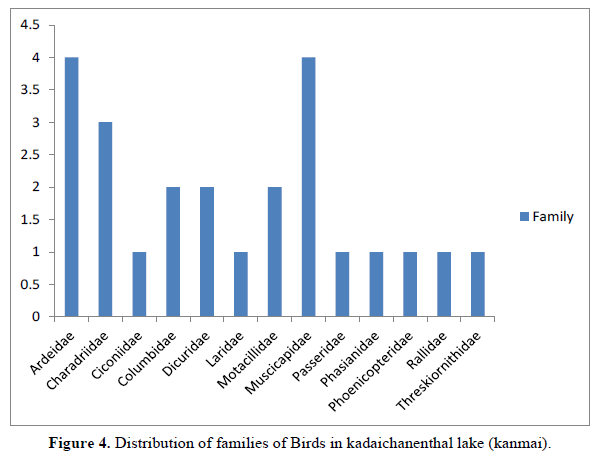
The most important threat to butterfly diversity is urbanization. Complete eradication of greenery in an area drives the butterfly population away since there is a lack of food and reduced chances to increase the progeny. Human activities have an undeniably strong influence on the biodiversity of all existing species. Due to the availability of varied sources of feed as well as foraging, the rich diversity of the birds were documented during the present study. The wetlands provides heterogeneous feeding habits to avifauna [12].Wetland habitat by supporting different food sources like planktons, fishes and invertebrates, further adding to the diversity of the wetlands [13]. The present study area of kadaichanenthal lake recorded of birds 24 species, 5 order and 13 families (Table 2). The family wise distribution stutes recorded families are Ardeidae (4 species), Charadriidae(3 species) Ciconiidae(1 species), Columbidae(2 Species), Muscicapidae (4species), Motacillidae (2 species), Dicuridae (2species), by following families are only one species recorded Laridae, Passeridae (1species), Phasianidae, Phoenicopteridae, Rallidae, Threskiornithidae (Figure 4).
| S.No | Common Name | Scientific name | Order | Family |
|---|---|---|---|---|
| 1 | Indian Peafowl | Pavo cristatus | Galliformes | Phasianidae |
| 2 | White-breasted Waterhen | Amaurornis phoenicurus | Gruiformes | Rallidae |
| 3 | Blue Rock Pigeon | Columba livia | Columbiformes | Columbidae |
| 4 | Little Brown Dove | Little Brown Dove | Columbiformes | Columbidae |
| 5 | Black Drongo | Dicrurus macrocercus | Passeriformes | Dicuridae |
| 6 | Ashy Drongo | Dicrurus leucophaeus | Passeriformes | Dicuridae |
| 7 | Red-breasted Flycatcher | Ficedula parva | Passeriformes | Muscicapidae |
| 8 | Tickell’s Blue Flycatcher | Cyornis tickelliae | Passeriformes | Muscicapidae |
| 9 | Oriental Magpie Robin | Copsychus saularis | Passeriformes | Muscicapidae |
| 10 | Indian Robin | Copsychus fulicatus | Passeriformes | Muscicapidae |
| 11 | House Sparrow | Passer domesticus | Passeriformes | Passeridae |
| 12 | Yellow Wagtail | Motacilla flava | Passeriformes | Motacillidae |
| 13 | White Wagtail | Motacilla alba | Passeriformes | Motacillidae |
| 14 | Grey Heron | Ardea cinerea | Ciconiiformes | Ardeidae |
| 15 | Pond Heron | Ardeola grayii | Ciconiiformes | Ardeidae |
| 16 | Cattle Egret | Bubulcus ibis | Ciconiiformes | Ardeidae |
| 17 | Large Egret | Ardae alba | Ciconiiformes | Ardeidae |
| 18 | Painted Stork | Mycteria leucocephala | Ciconiiformes | Ciconiidae |
| 19 | Black Ibis | Pseudibis papillosa | Ciconiiformes | Threskiornithidae |
| 20 | Flamingo | Phoenicopterus roseus | Ciconiiformes | Phoenicopteridae |
| 21 | Redwattled Lapwing | Vanellus indicus | Gruiformes | Charadriidae |
| 22 | Yellow-wattled Lapwing | Vanellus malabaricus | Gruiformes | Charadriidae |
| 23 | Little Ringed Plover | Charadrius dubius | Gruiformes | Charadriidae |
| 24 | Indian River Tern | Sterna aurantia | Gruiformes | Laridae |
Table 2. Avifauna of water lake (kanmai) kadaichanenthal at Madurai district, Tamil Nadu.
Many researchers done related work such as recorded Ardeidae to be the most dominant family in Bharathpuzha river basin in Kerala, and Surana recorded Anatidae to be the most dominant family in Chimdi lake Nepal [14]. Also the avifaunal diversity was studied by different authors from Maharashtra state such as recorded 78 species from Pohara- Malkhed forest reservoir of Amravati district, recorded 74 species from two freshwater lakes of Washim district, recorded 68 species from Chhatri lake of Amravati district, reported 72 species from Nagpur city, recorded 126 species from Navegaon national park from Gondia district, recorded 52 species from Shrungarbandh lake in Gondia district, recorded 131 species from Bhayander and Naigaon wetlands in Thane district, reported 27 species from Zaliya lake in Gondia district [15-20]. Durairaj reported 117 species from Thiruthalaiyur Lake Tiruchirapalli Forest Division, Tamil Nadu [21]. reported 51 species from Komaranahalli Lake, Davanagere District, Karnataka [22].
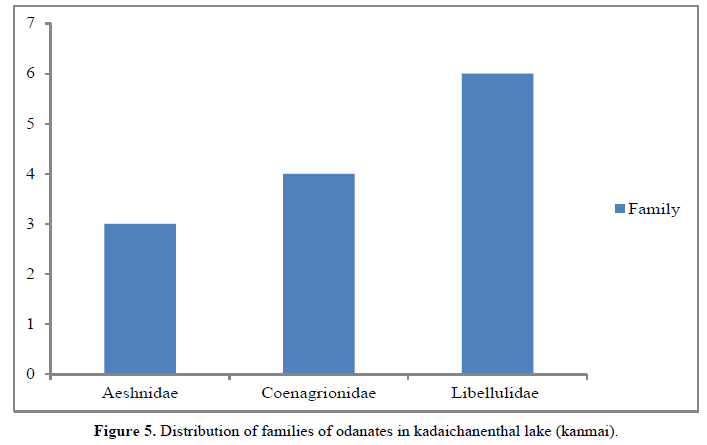
In present study reported in kadaichanenthal Lake Odanates are 13 species (Table 3). The family wise status are Aeshnidae (3 species), Coenagrionidae (4 species), Libellulidae (6 species), (Figure 5). Followed by 419 individuals under 4 families, 9 genera and 10 species of Odonata were recorded in Palni Hills of Western Ghats in tamilnadu. Among the Odonata families, Libellulidae was speciose (5 species) followed by Euphaeidae (2 species), Chlorocyphidae (1 species),Coenagrionidae (1 species) and Aeshnidae (1 species).
| S.No | Scientific name | Common name | Family |
|---|---|---|---|
| 1 | Brachythemis contaminata | Ditch jewel | Libellulidae |
| 2 | Crocothemis servilia | Ruddy Marsh Hawk | Libellulidae |
| 3 | Diplocodes trivialis | Ground Skimmer | Libellulidae |
| 4 | Pantala flavescens | Wandering Glider | Libellulidae |
| 5 | Urothemis signata | Greater Crimson Glider | Libellulidae |
| 6 | Rhyothemis variegata | Common picture wing | Libellulidae |
| 7 | Pseudagrion microcephalum | Blue river damsel | Coenagrionidae |
| 8 | Ischnura aurora | Golden darlet | Coenagrionidae |
| 9 | andering midget | Wandering midget | Coenagrionidae |
| 10 | Ischnura senegalensis | Marsh blue tail | Coenagrionidae |
| 11 | Anax guttatus | pale-spotted emperor | Aeshnidae |
| 12 | Gynacantha bayadera | parakeet darner | Aeshnidae |
| 13 | Anax immaculifrons | blue darner | Aeshnidae |
Table 3. Odonata of water lake (kanmai) kadaichanenthal at Madurai district, Tamil Nadu.
Environmental factors like water quality, altitude and nutrient supplements influenced the diversity of aquatic fauna and also showed great impact on species richness [23]. Several investigators have reported that dragonflies and damselflies are very common in rice agroecosystem. Kandibane have recorded 12 species of Odonata under three families in rice fields of Madurai, Tamil Nadu. More than 70 per cent species belonging to these families that occur in India are endemic to Western Ghats [24]. Libellulidae and Gomphidae are well-distributed Anisopteran members across Indian subcontinent, with few species restricted to Western Ghats and/or northeast India [25- 27]. It is opined that in place wherever deforestation, canalization of water bodies, urbanization, industrial development and disturbance of the natural cropping systems with agrochemicals was seen, dominance of Libellulidae was observed [28,29].
Conclusion
The study documents the rich avifauna shows that the area still provides some potential habitats for declining population of the species. Study area is situated near the city. So it has high human interference. Anthropogenic activities may be the reason for depressed species diversity. Unfortunately, these regions are getting invalid by commercial activity due to urbanization. There is a need to monitor this area systematically with focused study and a conservation plan should be under taken by the government to save the urban species and their sustainable production.
References
- Wanjari, P.D., 2012. Avifaunal diversity of Nagpur city, MS, India. Bionano Frontier., 5: 124-126.
- Alarape, A.A., Omifolaji, J.K., and Mwansat, G.S., 2015. Butterfly species diversity and abundance in University of Ibadan Botanical Garden, Nigeria. Open. J. Ecol., 5: 352.
- Gaonkar, H., 1996. Butterflies of Western Ghats, including Sri Lanka, A biodiversity assessment of a threatened mountain system. Nature., 32: 109-110.
- Grimmett, R., Inskipp, C., and Inskipp, T., 2011. Birds of the Indian subcontinent. Oxford University Press, 528.
- Islam, M.Z., and Rahmani, A.R., 2004. Important Bird Areas in India: priority sites for conservation. Indian Bird Conservation Network: Bombay Natural History Society and Birdlife International (UK), 1133.
- Surana, R., Subba, B.R., and Limbu, K.P., 2007. Avian diversity during rehabilitation stage of Chimdi Lake, Sunsari, Nepal. Our Nat., 5: 75-80.
- Siva, T., and Neelanarayanan, P., 2021. Diversity of Avifauna observed and recorded in Thinnanur Lake in Tiruchirappalli District, Tamil Nadu, India. Asian. J. Conserv. Biol., 10: 308-316.
- Ramachandra, T.V., 2006. Soil and groundwater pollution from agricultural activities. The Energy and Resources Institute (TERI).
- Subramanian K.A., 2014. A checklist of Odonata (Insecta) of India. Zoological Survey of India, Kolkata, West Bengal.
- McCoy, E.D., and Bell, S.S., 1991. Habitat structure: the evolution and diversification of a complex topic. Springer, 3-27.
- Hawkins, B.A., and Porter, E.E., 2003. Does herbivore diversity depend on plant diversity? The case of California butterflies. Am. Nat., 161: 40-49.
- Saikia, P.K., and Saikia, M.K., 2000. Diversity of bird fauna in NE India. J. Assam. Sci. Soc., 41: 379-396.
- Jules, E.S., and Dietsch, T.V., 1997. Dangers in dividing conservation biology and agroecology. Conserv. Biol., 11: 1272-1273.
- Kumar, A.B., 2006. A checklist of avifauna of the Bharathpuzha river basin, Kerala. Zoos. Print J., 21: 2300-2355.
- Kasambe, R., and Wadatkar, J., 2007. Birds of Pohara-Malkhed reserve forest, Amravati, Maharashtra-An updated annotated checklist. Zoos. Print J., 22: 2768-2770.
- Kedar, G.T., Patil, G.P., and Yeole, S.M., 2008. Comparative study of avifaunal status of two freshwater lakes of Washim district, Maharashtra. J. Aqua. Biol., 23: 29-33.
- Kukade, R.J., Warhekar, S.R., Tippat, S.K., and Dudhey, N.S., 2011. Avifaunal diversity of Chhatri lake, Amravati, Maharashtra. Biodivers. Conserv.
- SV, B., and Paliwal, G.T., 2014. Biodiversity and conservation status of water birds in Shrungarbandh lake district Gondia, Maharashtra, India. Int. J. of Life. Sciences., 2: 239-243.
- Lad, D., and Patil, S., 2015. Status and Diversity of Avian fauna in the estuarine wetland area of Bhayander and Naigaon, Maharashtra, India. Bio. Disc., 6: 39-44.
- Puri, S.D., 2015. Avifaunal Diversity of Malguzari Lake at Zaliya near Amgaon in Gondia district (MS) India. Int. J. of Life. Sciences., 3: 219-224.
- Durairaj, P., 2017. Study on avifaunal diversity from thiruthalaiyur lake Tiruchirapalli forest division, Tamil Nadu. J. Environ. Sci., Toxicol. Food. Technol., 12: 67-71.
- Harisha, M.N., and Hosetti, B.B., 2018. Status and conservation issues of wetland birds in Komaranahalli Lake, Davanagere District, Karnataka, India. J. Threat. Taxa., 10: 11290-11294.
- Pouilly, M., Barrera, S., and Rosales, C., 2006. Changes of taxonomic and trophic structure of fish assemblages along an environmental gradient in the Upper Beni watershed (Bolivia). J. Fish. Biol., 68: 137-156.
- Subramanian, K.A., 2007. Endemic odonates of the Western Ghats: habitat distribution and conservation. Odonata: Biol. Dragonflies., 257-271.
- Fraser, F.C., 1934. The Fauna of British India including Ceylon and Burma. Odonata Vol. II.Taylor and Francis Ltd., London.
- Fraser, F.C., 1936. The Fauna of British India including Ceylon and Burma. Odonata Vol. III.Taylor and Francis Ltd., London.
- Subramanian, K.A., 2005. Dragonflies and Damselflies of Peninsular India-A field Guide. Project Lifescape. Centre for Ecological Sciences.
- Subramanian, K.A., Ali, S., and Ramchandra, T.V., 2008. Odonata as indicators of riparian ecosystem health a case study from south western Karnataka, India. Fraseria., 7: 83-95.
- Subramanian, K.A., Kakkassery, F., and Nair, M.V., 2011. The status and distribution of dragonflies and damselflies (Odonata) of the Western Ghats. The status and distribution of freshwater biodiversity in the Western Ghats, India. International Union for Conservation Network and Zoo Outreach Organization, 63-72.
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref
Indexed at, Google Scholar, Cross Ref