Case Report - Current Trends in Cardiology (2020) Volume 4, Issue 1
An infrequent association: Left ventricular non compaction with bicuspid aortic valve and coarctation of the aorta.
Aureo Campos-Tello1*, Giancarlo A Valle1, Félix A Revilla1, Sara Ramírez-Flores2, Ángel Cueva- Parra1, Zoila Rodríguez-Urteaga3
1Department of Cardiology, Hospital Nacional Dos de Mayo, Lima, Peru
2Department of Cardiology, Hospital Central FAP, Lima, Peru
3Department of Cardiology, Instituto Nacional Cardiovascular INCOR, Lima, Peru
- Corresponding Author:
- Aureo Campos-Tello
Department of Cardiology
Hospital Nacional Dos de Mayo
Lima
Peru
E-mail: afernandocampos@gmail.com
Accepted date: Feb 20, 2020
Citation: Aureo Campos-Tello, Giancarlo A Valle, Félix A Revilla, et al. An infrequent association: Left ventricular non compaction with bicuspid aortic valve and coarctation of the aorta. Curr Trend Cardiol 2020;4(1):1-4.
DOI: 10.35841/cardiology.4.1.5-8
Visit for more related articles at Current Trends in CardiologyKeywords
Left ventricular non compaction, Bicuspid aortic valve, Coarctation of the aorta.
Introduction
Left ventricular non compaction (LVNC) is a rare congenital cardiomyopathy that can be diagnosed at any age. This disease makes the LV to become “spongy” and have two separate layers: a compacted epicardial layer and a large non compacted endocardial layer with trabeculation and deep recesses communicated within them but not with coronary circulation. Its reported prevalence is between 0.014 and 1.3% according to echocardiographic studies [1].
Despite the exact mechanisms are not well comprehended, several gene mutations (especially in MYH7 and MYBPC3 genes) avoid myocardium to become smooth and firm (compacted) during intrauterine life [2].
This condition is usually asymptomatic, but can be complicated by heart failure, thromboembolism or ventricular arrhythmias, including sudden cardiac death. It’s primarily diagnosed with echocardiography and confirmed with CMR [3].
In this manuscript, we report a case of LVNC associated with two other abnormalities: bicuspid aortic valve and coarctation of the aorta. This case is important because this congenital disease is underdiagnosed and its prompt recognition is necessary to avoid further complications.
Case Presentation
An asymptomatic 29-year-old male from Lima attended to a pre surgical evaluation for knee intervention, in our department. At physical exam, his vitals were: heart rate of 63 bpm, BP of 145/91 mmHg (measured in right arm), respiratory rate 14 breath/minute and blood oxygen saturation of 96%. The cardiovascular exam revealed a diastolic murmur in aortic area and diminished pulse amplitude in lower limbs. Therefore, BP was measured in left leg finding 100/60 mmHg. Due to described murmur and BP difference between arms and legs, there was suspicion of aortic regurgitation and coarctation of the aorta.
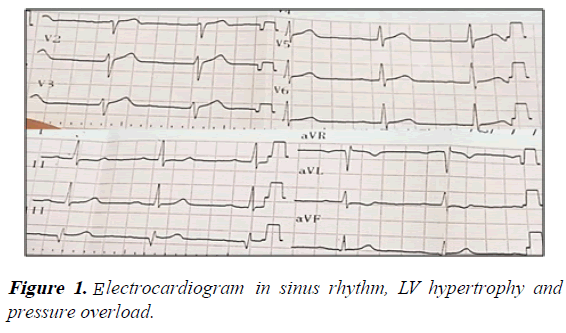
Electrocardiography showed sinus rhythm, normal axis, signs of LV hypertrophy and pressure overload (Figure 1).
The transthoracic echocardiography (TTE) showed the following findings:
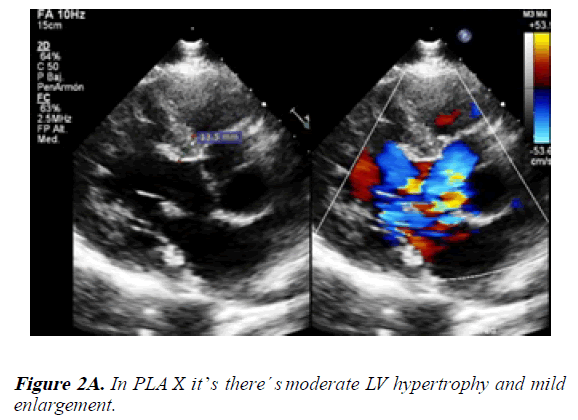
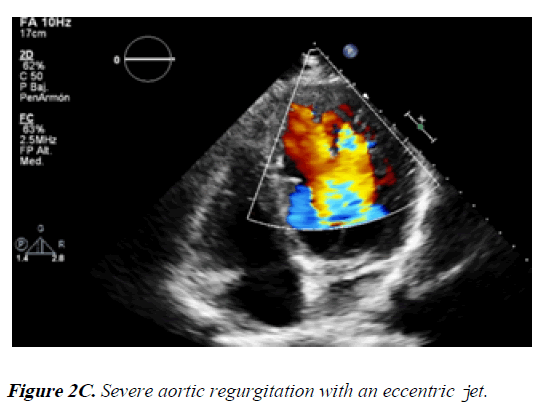
1) In the PLAX view we identified LV with mild enlargement and moderate hypertrophy (end diastolic diameter: 57.5 mm and end systolic diameter: 37.2 mm, IVSd: 13.5 mm). The aorta was dilated at the sinuses of Valsalva level (40.6 mm). Also, the aortic valve had severe regurgitation with an eccentric jet (Figures 2A and 2B).
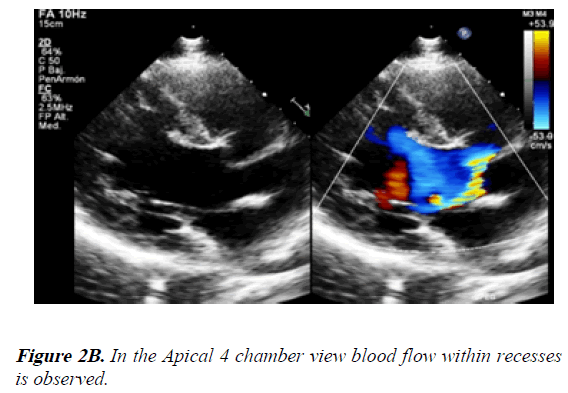
2) In the apical view it was possible to see blood flow within deep trabeculae and recesses (Figure 2C).
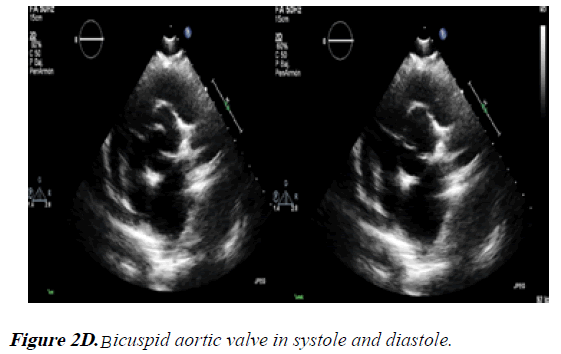
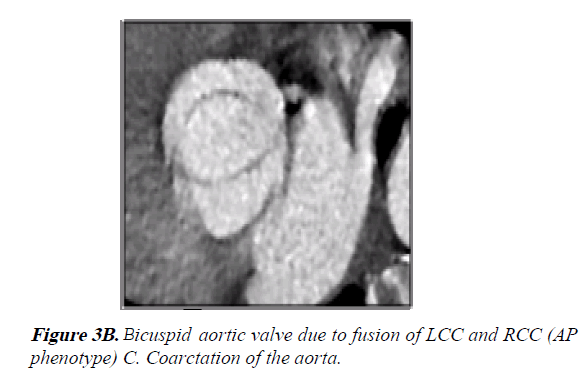
3) In the PSAX view the aortic valve seemed to be bicuspid due to fusion of LCC and RCC (anterior cusp in an AP phenotype). The NCC became the posterior cusp (Figure 2D).
4) In the suprasternal view, there was a narrowing in the descending thoracic aorta suggesting coarctation of the aorta. The CT confirmed the previous findings:
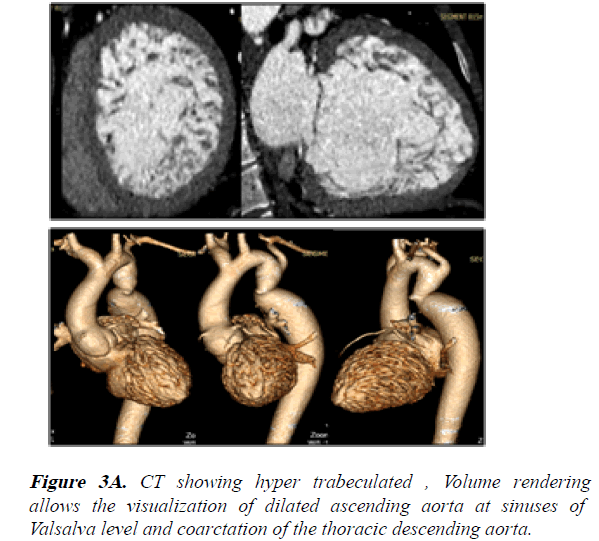
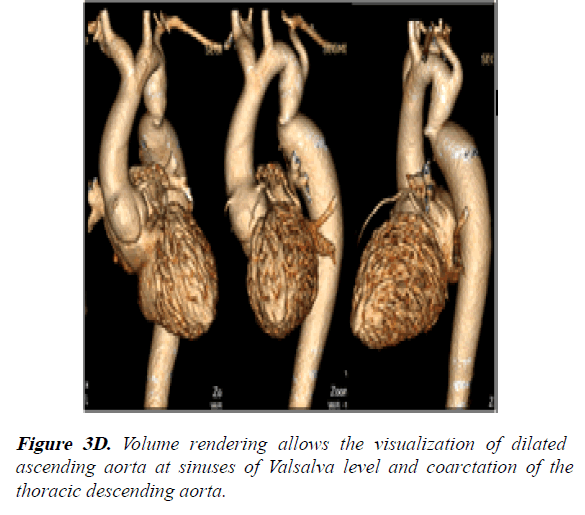
1) Hyper trabeculated LV and mild dilation of aorta at the sinuses of Valsalva level (42 mm) (Figures 3A-3D).
2) Bicuspid aortic valve (BAV) AP phenotype with fusion of LCC and RCC through a raphe (Figure 3B).
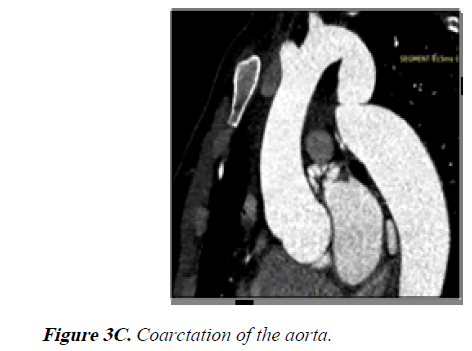
3) The coarctation of the aorta measured 14 × 19.5 mm with a 38% narrowing relative to the aortic diameter at the diaphragm level (Figures 3C and 3D).
The CMR allowed us to make our final diagnosis because:
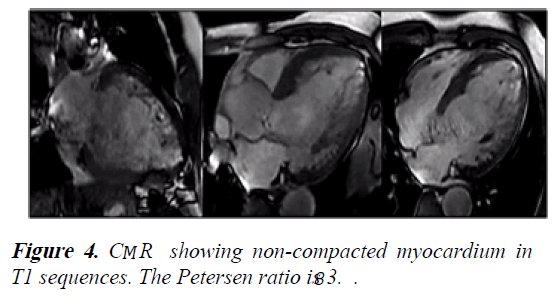
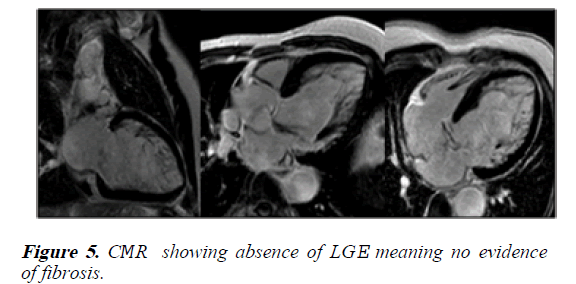
1) The ratio of noncompacted myocardium to compacted myocardium was 3.8. However, there was no LGE (Figures 4 and 5).
2) The LV ejection fraction was 65%, its EDV was 277 ml/m2 (indexed 164 ml/m2) and ESV was 97 ml/m2 (indexed: 58 ml/m2).
3) The maximum narrowing of the aorta at the coarctation was 12 mm.
After all the described workup process we concluded: left ventricular non compaction associated with bicuspid aortic valve and coarctation of the aorta.
We performed an exercise test (Bruce protocol) and the patient reached 7 METS. It was stopped because he had knee pain.
Discussion
Left ventricular non compaction is an entity recognized for 80 years and is considered by European Society of Cardiology as a not classified familiar cardiomyopathy. Its presentation is isolated or associated with another abnormalities [1]. There are few reported cases associated with bicuspid aortic valve and coarctation of the aorta.
There are several criteria for diagnosis depending on the method used for that purpose. However, CMR has the best performance. Jacquier et al. described criteria for diagnosis of this disease: a trabecular LV mass above 20% of total mass has a sensitivity of 91.6% and a specificity of 86.5% [4]. Nevertheless, the most used criteria for diagnosis by CMR is the one described by Petersen et al.: the ratio of noncompacted myocardium to compacted myocardium must be greater than 2.3 during diastole [5]. Therefore, our patient met the criteria for diagnosis of LVNC because he had a Petersen ratio of 3.8.
In current literature, there are some cases of LVNC with Ebstein’s anomaly, but there are few associated with bicuspid aortic valve or coarctation of aorta. The first one (LVNC and BAV) was reported in 2003 when Cavusoglu et al. described a 19 year-old-male with heart failure due to a severely enlarged LV with moderate impairment in systolic function (LVEF: 35%)[6]. In 2009, Wrigley et al. published another case of a 39 year-old-male with LV, BAV and aortic regurgitation, but the first one to undergo valve replacement surgery. He had a mildly dilated LV (end-diastolic diameter: 61 mm) and preserved systolic function, so he was initially medically managed. After 4 months of follow up, he had aortic valve replacement because of AR severity progression without variations in LV diameter or ejection fraction. The outcome was favorable [7].
An even more rare association is the one described for the first time by Myers et al.: a 15-year-old female with LVNC, BAV and coarctation of the aorta. She also had a subaortic VSD. Thus, the LV had severely enlargement and impairment of ejection fraction. This complex was called by the authors as Laubry-Pezzi syndrome [8].
Our patient had the association of LVNC, bicuspid aortic valve with severe regurgitation and coarctation of the aorta. None of the above cases mentioned before had a patient with these three features. Despite the severity of aortic regurgitation and mild LV enlargement (measured by TTE and CMR), the patient had a NYHA I functional class with preserved LVEF; thus, we decided medical therapy. This conservative approach was the same applied initially by Wrigley et al. [7].
Even in presence of bicuspid aortic valve and severe aortic regurgitation, we decided for a conservative strategy because, according to the 2017 European Society of Cardiology Guidelines for the management of valvular heart disease, a patient with severe AR should undergo surgery if symptomatic, LVEF<50%, LVEDD>70 mm or LVESD>50 mm); our patient didn’t met none of this criteria. It’s also known that a patient with severe AR should undergo surgery if it’s planned an intervention of ascending aorta, but our patient didn’t have that indication either. This happens because in presence of bicuspid aortic valve, a patient should be scheduled for surgery if the maximal ascending aortic diameter ≥50 mm with risk factor or ≥55 mm without them; our patient didn’t reach those cutoffs (he had 42 mm at the sinuses of Valsalva level) [9].
Finally, the coarctation of the aorta had class I indication for intervention due to BP>140/90 mmHg (145/91 mmHg), BP difference>20 mmHg between upper and lower limbs (in our case was 45 mmHg) and significant LV hypertrophy detected by TTE (IVSd: 13.5 mm) [10]. Therefore, we decided for percutaneous intervention of the coarctation, medical therapy and clinical-imaging follow up. The patient received Enalapril 5 mg PO BID and Furosemide 40 mg PO QD. By now, he continues with that treatment, remains intervention.
Conclusion
The patient has certain features not reported before in current literature. That’s why we decided to publish this case. We’re completely sure that if this patient only had LVNC, he’ll never had his disease detected because most of LVNC are asymptomatic. The associated abnormalities (BAV with AR and coarctation of the aorta) made possible to identify and diagnose this patient, since it all started with just a murmur. We should emphasize the importance of an accurate physical examination and non-invasive images that that played a crucial role in this case.
References
- Feltes-Guzmán G, Nunez-Gil I. Left ventricular non compaction. An article from the e-journal of the ESC Council for Cardiology Practice. European Society of Cardiology. 2012; 10(31):26.
- Dong X, Fan P, Tian T, et al. Recent advancements in the molecular genetics of left ventricular noncompaction cardiomyopathy. Clin Chim Acta. 2017; 465:40-44.
- Finsterer J, Stöllberger C, Towbin J. Left ventricular non compaction cardiomyopathy: Cardiac neuromuscular, and genetic factors. Nature Reviews Cardiology.2017; 14(4):224-37.
- Jacquier T, Thuny F, Jop B, et al. Measurement of trabeculated left ventricular mass using cardiac magnetic resonance imaging in the diagnosis of left ventricular non-compaction. European Heart Journal.2010; 31(9):1098-1104.
- Petersen SE, Selvanayagam JB, Wiesmann F, et al. Left ventricular non-compaction: Insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol. 2005; 46 (1):101-05.
- Cavusoglu Y, Ata N, Timuralp B, et al. Noncompaction of the ventricular myocardium: Report of two cases with bicuspid aortic valve demonstrating poor prognosis and with prominent right ventricular involvement. Echocardiography. 2003; 20(4):379-383.
- Wrigley Benjamin J, Rosin M, Banerjee P. Replacement of a congenital bicuspid aortic valve in a patient with left ventricular noncompaction. Tex Heart Inst J. 2009; 36(3):241-43.
- Myers PO, Tissot C, Cikirikcioglu M, et al. Complex aortic coarctation, regurgitant bicuspid aortic valve with VSD and ventricular non-compaction: A challenging combination. Thorac Cardiov Surg. 2011; 59(5):313-16.
- Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017; 38(36):2739-91.
- Baumgartner H, Bonhoeffer P, De Groot NM, et al. ESC Guidelines for the management of grown-up congenital heart disease. Eur Heart J. 2010; 31(23):2915-57.