Case Report - Journal of Orthopedic Surgery and Rehabilitation (2017) Volume 1, Issue 2
Palmar fasciectomy after collagenase Clostridium histolyticum injection for Dupuytrens contracture: A case report.
Michiro Yamamoto*, Takanobu Nishizuka and Hitoshi Hirata
Department of Hand Surgery, Nagoya University Graduate School of Medicine, Japan
- *Corresponding Author:
- Michiro Yamamoto
Department of Hand Surgery Nagoya University Graduate School of Medicine 65 Tsuruma-cho, Showa-ku, Nagoya, 466-8550 Japan
E-mail: michi-ya@med.nagpya-u.ac.jp
Accepted date: May 16, 2017
Citation: Yamamoto M, Nishizuka T, Hirata H. Palmar fasciectomy after collagenase Clostridium histolyticum injection for Dupuytren’s contracture: A case report. . J Orthop Surg Rehabil 2017;1(2):1-4.
Abstract
We present a case of successful salvage fasciectomy after initial collagenase Clostridium histolyticum (CCH) injection for the treatment of Dupuytren’s contracture. We did not find any remarkable scarring around the Dupuytren’s cord and nodule three and a half years after the initial CCH injection. Pathological findings revealed the previously ruptured cord and partial continuity of the collagen tissue. If there is a sufficient duration between initial CCH injection and salvage surgery, fasciectomy can be performed without an adverse effect of the enzyme around the Dupuytren’s cord and nodule.
Keywords
Dupuytren's contracture, Collagenase clostridium histolyticum injection, Palmar fasciectomy.
Introduction
Dupuytren’s contracture (DC) is a fibroproliferative disease of the palmar fascia with unknown etiology [1]. Abnormal fibroblasts and myofibroblasts of the palmar fascia have been implicated in disease progression [1]. DC tends to worsen and progress with age, resulting in compromised hand function due to finger contractures. Treatment options for contractures include cutting of the Dupuytren’s cord (fasciotomy) or surgical resection (fasciectomy) [1]. Recently, collagenase Clostridium histolyticum (CCH) injection has been approved by the Food and Drug Administration (FDA) in the United States following the results of a double blind, placebocontrolled, multicenter trial that demonstrated its effectiveness [2]. In 2015, CCH was also approved by the Ministry of Health, Labour and Welfare in Japan [3].
Although CCH injection is a less invasive nonsurgical treatment option for DC, it is not without its associated complications. In fact, the complication rate of CCH injection was comparable with that of surgery in one study [4]. Flexor tendon rupture is one of the most serious complications of CCH injection with an incidence of 0.3% [4]. Less severe complications include subcutaneous inflammation and secondary scarring. In a case report of flexor tendon rupture due to CCH injection, second stage reconstruction with a tendon graft was performed because of extensive soft tissue inflammation around the ruptured flexor tendons [5]. Another limitation of CCH injection is recurrence. In one study that followed patients after CCH injection with more than 20 degrees of flexion contracture, 47% (291 of 623) of cases recurred at 5 years [6]. Salvage fasciectomy after initial treatment with CCH injection may be a suitable management option, but this surgical technique requires a skilled operator since the inflammatory cascade will be stimulated around the injected site, and secondary subcutaneous scarring may be present.
We report herein a case of salvage fasciectomy at three and a half years after initial CCH injection with pathological findings of resected Dupuytren’s cord and nodule.
Case Report
This retrospective case report did not require the approval of the institutional review board. The patient gave informed consent for his anonymized data to be published.
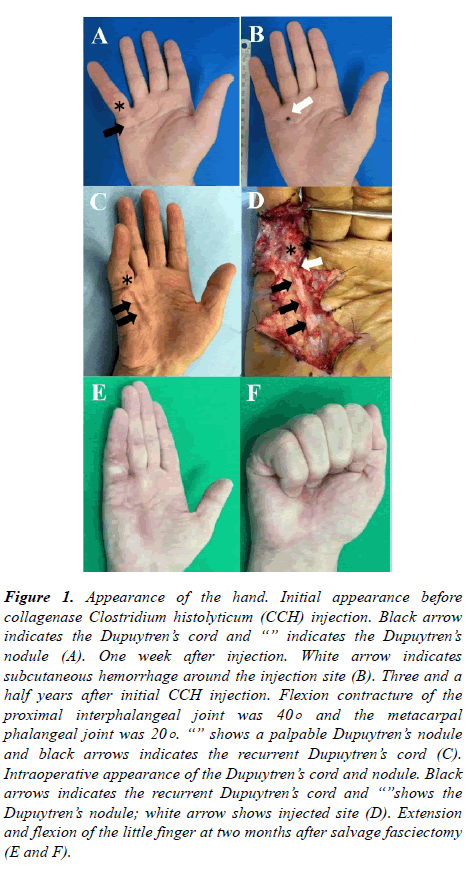
A 69-year-old man presented with a history of progressive right little finger contracture. He first noticed the painless nodule on the ulnar side of the right palm five years prior to presentation at our hospital. His past medical history was significant for lip cancer that was surgically treated and in remission. Physical examination revealed 45-degree flexion contracture of the metacarpophalangeal (MP) joint of the little finger with a palpable pretendinous cord and Dupuytren’s nodule on the volar side of MP joint (Figure 1A).
Figure 1. Appearance of the hand. Initial appearance before collagenase Clostridium histolyticum (CCH) injection. Black arrow indicates the Dupuytren’s cord and “” indicates the Dupuytren’s nodule (A). One week after injection. White arrow indicates subcutaneous hemorrhage around the injection site (B). Three and a half years after initial CCH injection. Flexion contracture of the proximal interphalangeal joint was 40° and the metacarpal phalangeal joint was 20° . “” shows a palpable Dupuytren’s nodule and black arrows indicates the recurrent Dupuytren’s cord (C). Intraoperative appearance of the Dupuytren’s cord and nodule. Black arrows indicates the recurrent Dupuytren’s cord and “”shows the Dupuytren’s nodule; white arrow shows injected site (D). Extension and flexion of the little finger at two months after salvage fasciectomy (E and F).
Since the patient suffered from contracture of the hand, we discussed treatment options including fasciectomy and CCH injection, and he selected the latter. CCH (0.58 mg) was injected into the Dupuytren’s cord located 1 cm proximal to the MP joint of the little finger, and the finger was successfully manipulated the day after injection.
The patient was reviewed at 7 days (Figure 1B), 30 days, and 3, 6, and 12 months after the procedure; there were not significant complications. After one year, he was monitored every six months.
One and a half years after initial CCH injection, he developed flexion contracture of the proximal interphalangeal joint of the right little finger, and this progressed gradually up to 40 degrees of extension deficit. Three years after initial CCH injection, flexion contracture of the MP joint of the same finger occurred and progressed up to 20 degrees of extension deficit, and his right hand function became limited due to these contractures. He was diagnosed with grade 3 contracture of his right hand as defined by the Meyerding classification (Figure 1C) [7]. We re-discussed treatment options with the patient, and he selected surgical fasciectomy.
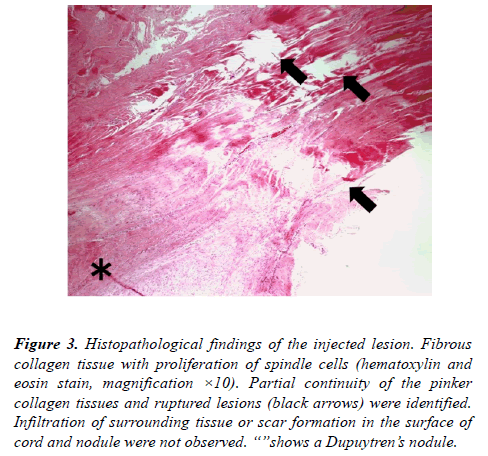
We performed fasciectomy under axial block with air tourniquet control. We made a zigzag incision over the Dupuytren’s cord using a magnifying glass. Before surgery, we imaged the scar formation around the CCH injection site, but did not notice any differences in the subcutaneous tissue compared to the primary fasciectomy (Figure 1D). We performed subtotal resection of the affected palmar fascia including the pretendinous cord and Dupuytren’s nodule without any more difficulty than expected with primary fasciectomy. We could not find any additional scarring around the Dupuytren’s cord and nodule. Macroscopically, the Dupuytren’s cord was completely regenerated and had recurred (Figure 2); however, microscopically, the proximal and distal part of the Dupuytren’s cord were connected incompletely. Pathological examination of the removed Dupuytren’s cord and nodule revealed fascia with infiltrates of spindle cells and a previously ruptured cord with partial continuity of the pinker collagen tissues (Figure 3). We could not find any infiltration of the surrounding tissue or scar formation in the outer part of the cord and nodule.
Figure 3. Histopathological findings of the injected lesion. Fibrous collagen tissue with proliferation of spindle cells (hematoxylin and eosin stain, magnification ×10). Partial continuity of the pinker collagen tissues and ruptured lesions (black arrows) were identified. Infiltration of surrounding tissue or scar formation in the surface of cord and nodule were not observed. “”shows a Dupuytren’s nodule.
The patient started active finger motion exercises from the second postoperative day, and suture strings were removed three weeks after fasciectomy. He resumed his daily activities without loss of motion or any residual symptoms (Figure 1E and F). One year after fasciectomy, extension degrees of the right little finger were 20 and -15 for MP and PIP joints, respectively. There was no recurrence of the cord and symptoms.
Discussion
Five-year recurrence rates after CCH injection for joints with > 20° contracture were 39% in the MP joint and 66% in the PIP joint [6]. These rates are superior to the 5-year recurrence rates after surgical fasciotomy (85%) and worse than those following fasciectomy (21%) [8]. Given the increasing use of CCH injection for DC, it is likely that the use of salvage treatments as described in this case will also increase. Although the patient in this report opted for fasciectomy as the salvage procedure, he could have received another CCH injection. Salvage with CCH injection can be applied after both initial CCH injection and fasciectomy. However, there is limited evidence regarding the outcome of salvage fasciectomy after initial CCH injection. Everlin et al. reported the outcomes of 7 MP and 12 PIP joints following salvage fasciectomy after CCH injection [9]. The mean interval between the last CCH injection and salvage fasciectomy was 12 months. The authors found disruption of normal architecture and areolar tissue with extensive scarring in the injected lesion. The mean numbers of preoperative flexion contractures for the MP and PIP joints were 42 and 60 degrees, respectively. After salvage surgery, the mean degrees of the contractures were 0 and 21 for MP and PIP joints, respectively. Although CCH injection might cause a deeply scarred bed and increase surgical difficulty, results after salvage fasciectomy were favorable and comparable to those following primary fasciectomy [9]. Hay et al. reported a survey on the surgical difficulty after initial CCH injection [10]. Seven of nine surgeons reported that there was no significant distortion of anatomy and ranked the technical difficulty level as equivalent to a primary fasciectomy. In one of 15 cases, wound dehiscence occurred [10]. The histopathological findings after CCH injection have not been extensively reported. Sanjuan-Cervero et al. identified a completely normal cord on pathology after salvage fasciectomy [11]. Although the Dupuytren’s cord in our case appeared macroscopically normal, microscopic examination revealed incomplete continuity of the collagen tissues. This effect is secondary to the collagenase enzyme, which lasts approximately 12 hours [12]. However, after CCH injection, the pathology of the recurrent Dupuytren’s cord differed from the primary cord even three and a half years after initial CCH injection.
The presented case had a palpable Dupuytren’s nodule on the volar side of proximal phalanx. Although the primary affected lesion in our patient was the MP joint, the flexion contracture recurred in the PIP joint of the little finger. Enzymatic injection with CCH can degrade the collagen triple helix of the Dupuytren’s cord without any structural damage to noncollagenous tissue elements, [12] however, it cannot remove the entire Dupuytren’s nodule. The Dupuytren’s nodule on the volar side of the proximal phalanx with highly cellularity might have been the supply source of abnormal fibroblasts and myofibroblasts, which rebuilt the Dupuytren’s cord and resulted in flexion contracture of both the PIP and MP joints.
The outcome of salvage fasciectomy is influenced by numerous factors including the number of CCH injections into the joint, the duration between surgery and injection, and preoperative severity of the contracture. Although the presented case had Meyerding Grade 3 contracture, a minimal number of CCH injections and long interval of three and a half years after initial injection contributed to the patient’s favorable outcome.
Conclusion
In conclusion, we present a case of successful salvage fasciectomy after initial CCH injection for the treatment of Dupuytren’s contracture. We could not find any remarkable scarring around the Dupuytren’s cord and nodule three and a half years after the initial CCH injection. Pathological findings revealed the previously ruptured cord and partial continuity of the collagen tissue. If there is a sufficient duration between initial CCH injection and salvage surgery, fasciectomy can be performed without an adverse effect of the enzyme around the Dupuytren’s cord and nodule. Notably, the patient described in this case resumed his daily activities and did not suffer any complications after salvage fasciectomy, thus demonstrating the safety and efficacy of this procedure.
References
- Shih B, Bayat A. Scientific understanding and clinical management of Dupuytren disease. Nat Rev Rheumatol 2010; 6: 715-26.
- Hurst LC, Badalamente MA, Hentz VR, et al. Injectable collagenase clostridium histolyticum for Dupuytren's contracture. N Engl J Med 2009; 361: 968-79.
- Hirata H, Tanaka K, Sakai A, et al. Efficacy and safety of collagenase Clostridium histolyticum injection for Dupuytren's contracture in non-Caucasian Japanese patients (CORD-J Study): The first clinical trial in a non-Caucasian population. J Hand Surg Eur Vol 2016.
- Peimer CA, Wilbrand S, Gerber RA, et al. Safety and tolerability of collagenase Clostridium histolyticum and fasciectomy for Dupuytren's contracture. J Hand Surg Eur Vol 2015; 40: 141-49.
- Zhang AY, Curtin CM, Hentz VR. Flexor tendon rupture after collagenase injection for Dupuytren contracture: A case report. J Hand Surg Am 2011; 36: 1323-325.
- Peimer CA, Blazar P, Coleman S, et al. Dupuytren contracture recurrence following treatment with collagenase clostridium histolyticum (CORDLESS [Collagenase Option for Reduction of Dupuytren Long-Term Evaluation of Safety Study]): A 5-year data. J Hand Surg 2015; 40: 1597-605.
- Meyerding HW. Dupuytren's contracture. Arch Surg 1936; 32: 320-33.
- Van Rijssen AL, Ter Linden H, Werker PM. Five-year results of a randomized clinical trial on treatment in Dupuytren's disease: percutaneous needle fasciotomy versus limited fasciectomy. Plast Reconstr Surg 2012; 129: 469-77.
- Eberlin KR, Kobraei EM, Nyame TT, et al. Salvage palmar fasciectomy after initial treatment with collagenase clostridium histolyticum. Plast Reconstr Surg 2015; 135: 1000e-6e.
- Hay DC, Louie DL, Earp BE, et al. Surgical findings in the treatment of Dupuytren's disease after initial treatment with clostridial collagenase (Xiaflex). J Hand Surg Eur Vol 2014; 39: 463-65.
- Sanjuan-Cervero R, Vaquero M, Carrera-Hueso FJ. Salvage palmar fasciectomy after initial treatment with collagenase Clostridium histolyticum. Plast Reconstr Surg 2016; 137: 760e-1e.
- Del Carlo M, Cole AA, Hart SGE, et al. Comparative analysis of collagen degradation in Peyronie's disease plaque and Dupuytren's contracture cord tissues injected with mixed collagenase subtypes. J Urology 2009; 181: 279.